TWIN-COLUMN ENRICHMENT
Continuous Enrichment Chromatography with N-Rich®
Automated Trace Impurity Isolation. Accumulate what’s barely there — cyclic enrichment for reference standard preparation, charge variant isolation, and ICH Q3A/Q6B characterization: 100–1,000× Enrichment factor | 80–95% Purity overnight | fully automated via ChromIQ® N-Rich® Wizard.
Sources: Bigelow et al. (BMS), J. Chromatogr. A 2021; Weldon & Müller-Späth, J. Chromatogr. A 2022

The Isolation Bottleneck
Regulatory agencies require pure, isolated impurity reference standards — not just detected peaks. ICH Q3A and Q6B mandate structural identification and toxicological qualification of trace impurities and product-related variants. The problem is that these compounds exist at 0.1–5% of the feed, co-eluting with a dominant main peak that is 10–1000× more abundant.
Conventional batch preparative HPLC has no mechanism to address this ratio. Hundreds of injections, progressive pooling of dilute fractions, weeks of processing time — and at the end of it, material that may still be too impure to use as a reference standard. Custom synthesis costs tens of thousands of dollars per compound and is simply impossible for biologic impurities such as charge variants, glycoforms, or partially filled capsids that only exist as product-related species.

N-Rich® Changes What’s Possible
N-Rich® does not try to isolate a trace compound in a single pass. Instead, it accumulates it on-column across many cycles — progressively building concentration in the system until enough pure material is available to collect in a single, fine-fractionated elution.
Each enrichment cycle adds more of the target to the column while simultaneously removing the dominant main compound to waste. After enough cycles, the target has been concentrated to the point where it can be eluted and fractionated with purity that would be impossible at the original abundance. The process runs fully automated and unattended — overnight, or over multiple days for very low-abundance targets.
The results across validated applications are consistent:
95% purity
mAb charge variants in 10h vs. 300h by HPLC (BMS)
3 days
vs. 30+ days biosimilar mAb, 9 isoforms
79×
Productivity gain peptide impurity isolation
69×
Solvent reduction vs. analytical HPLC
Sources: Bigelow et al. (BMS), J. Chromatogr. A 2021 · Weldon & Müller-Späth, J. Chromatogr. A 2022
N-Rich® is a fundamentally different tool from MCSGP. MCSGP purifies a main product by removing flanking impurities — its starting material is already relatively pure. N-Rich® accumulates toward a compound that starts at trace abundance and may never be resolvable in a single-column run. Both use the same twin-column platform and similar ChromIQ® wizard structure; the problem they solve is different.
How N-Rich® Works…. Part 1
N-Rich® runs as a single uninterrupted procedure through four sequential stages:
| Stage | What happens | Duration |
|---|---|---|
| Design | Batch chromatogram loaded, recycling boundaries set, method generated | One-time setup |
| Enrichment | n cycles of cyclic accumulation — target compound builds on-column, main compound removed each cycle | n cycles until sufficient feed is processed |
| Depletion | One switch without fresh feed — remaining main compound removed, target concentrated before elution | 1 switch |
| Fractionated Elution | Shallow gradient across both columns in series — target eluted and fine-fractionated for collection | Single elution event |
How N-Rich® Works…. Part 2
Within each enrichment cycle, two switches alternate. Each switch consists of four phases running on the upstream (eluting) and downstream (regenerating/loading) columns in parallel:
| Phase | Upstream column | Downstream column |
|---|---|---|
| P1 – Non-target elution (Parallel) | Weakly adsorbing non-targets eluted to waste | Regenerated and re-equilibrated |
| P2 – Weak (W) recycling (Interconnected) | Weakly adsorbing target compounds transferred with in-line dilution to downstream column | Receives W fraction — target re-adsorbs |
| P3 – Main compound removal (Parallel) | Non-target (main) compound eluted to waste | Loaded with fresh feed — more target enters system |
| P4 – Strong (S) recycling (Interconnected) | Strongly adsorbing target compounds transferred with in-line dilution to downstream column | Receives S fraction — target re-adsorbs |

How N-Rich® Works…. Part 3
At the end of each switch, the two columns swap positions by valve switching and the cycle repeats. The feed volume per cycle is calculated automatically by the N-Rich Wizard from the batch chromatogram.
The Depletion stage (one switch, no fresh feed) transfers any remaining enriched material from the upstream to the downstream column before elution, minimizing non-target compound in the final fractions and improving final purity.
The Fractionated Elution uses a shallow gradient run across both columns in series — at half the normal flow rate with a flatter gradient than the batch method — to maximize resolution between enriched compounds. This is where the concentrated, pre-enriched target material is collected as fine fractions.

AutoPeak® — Product Removal Control
N-Rich’s AutoPeak® implementation is distinct from MCSGP’s. In MCSGP, AutoPeak® controls up to four fraction collection boundaries around a main product peak. In N-Rich®, AutoPeak® controls a single transition: when to start removing the main compound during the enrichment phase.
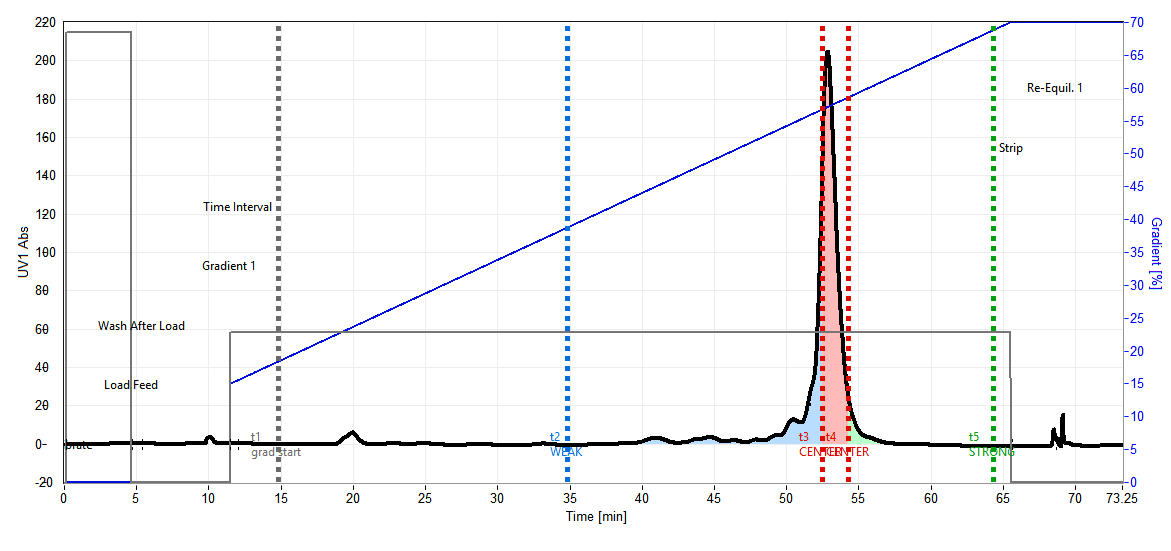
During each switch, the main (non-target) compound must be actively diverted to waste at the right moment to prevent it from accumulating. With AutoPeak® off, this diversion happens at fixed times t3 and t4 derived from the batch chromatogram. Fixed timing is appropriate during early method development with stable, well-characterised conditions.
With AutoPeak® on, the start of the removal is UV-triggered. If the removal boundary (t3) falls on the ascending slope of the main compound peak, removal is triggered by an absolute mAU threshold (Front Control). If t3 falls on the descending slope, removal is triggered by a relative threshold as a percentage of the peak maximum (Tail Control). In both cases, removal stops after a fixed time window (t4 − t3) — the stop is time-based, not UV-triggered.
A Threshold Detection Window acts as a safety margin: if the UV trigger is not reached within this extended window, the process freezes before the next phase rather than proceeding blindly — ensuring accumulated material is never compromised by a missed trigger during unattended multi-day runs.
Verifying Results: The Evaluation Center
Every N-Rich® run is captured in the Evaluation Center — ChromIQ®’s integrated data analysis module.
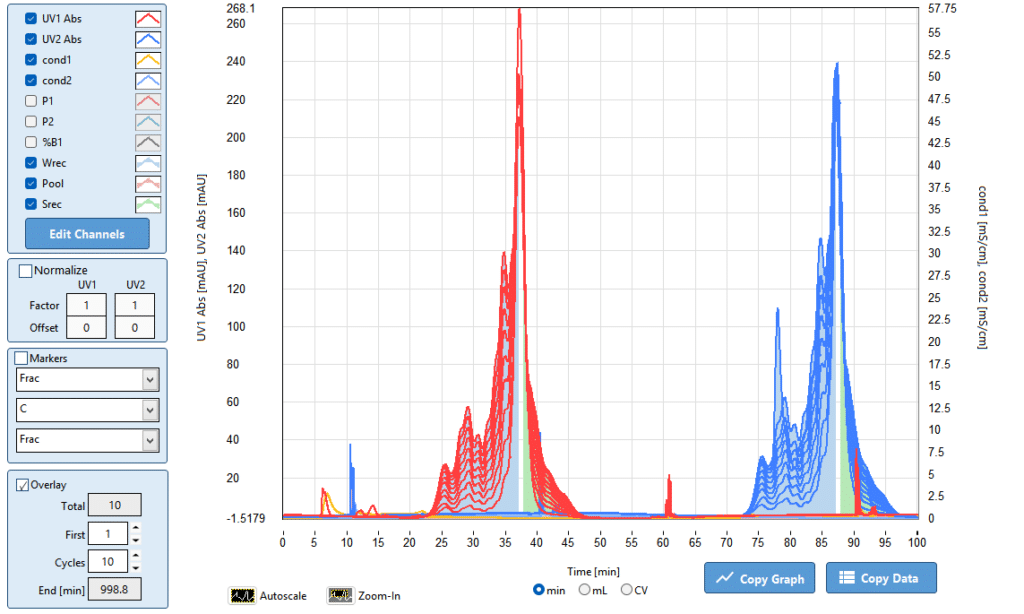
- Cycle Overlay — Superimpose all enrichment cycles in one view. The target compound UV signal should grow progressively taller across cycles while the main compound signal diminishes — visual confirmation that accumulation is proceeding as designed
- Auto Integration — Quantitatively tracks the enriched fraction areas across every cycle, confirming the rate of accumulation and documenting when the enrichment plateau has been reached
- Superimpose — Overlay the batch design chromatogram against the final Fractionated Elution output — the side-by-side comparison shows directly how much the target compound has been enriched relative to the main compound
- PDF and Excel export — Per-cycle chromatograms, Auto Integration results, and fractionation tables compiled into a structured report
Platform Advantages at a Glance
| Advantage | What it means in practice |
|---|---|
| 100–1,000× enrichment factor | Trace compounds at 0.1–5% of feed concentrated to high purity in a single automated run |
| Up to 80× faster than analytical HPLC | Hours or days instead of weeks — overnight operation for many applications |
| 3 days vs. 30+ days | Biosimilar mAb isoform isolation: complete in 3 days, 50 analytical samples vs. >600 |
| 95% purity — 30× faster than HPLC | mAb charge variants: 95% purity in 10 hours vs. 300 hours by analytical HPLC |
| Up to 69× solvent reduction | Same enrichment result with dramatically lower mobile phase consumption and waste |
| Same resin and buffers | No change to stationary phase or mobile phase conditions — your existing batch conditions are the starting point |
| Fully unattended | Procedure runs overnight or over multiple days without operator supervision — AutoPeak® maintains consistent depletion |
| Scale-up path | Methods developed on the Contichrom® CUBE transfer to the Contichrom® PILOT 300X and TWIN HPLC for higher throughput |
N-Rich®Applications
N-Rich® is applicable wherever a target compound exists at low abundance relative to a dominant co-eluting species and cannot be isolated to useful purity or quantity by conventional batch methods.
| Molecule class | Typical target | Use case |
|---|---|---|
| Monoclonal antibodies | Acidic and basic charge variants (typically 5–25% of feed) | Reference standards for forced degradation studies, biosimilar comparability, potency assay development |
| Biosimilar mAbs | Multiple isoforms from 1.5% to 56% of feed | Complete isoform panel in 3 days — all main isoforms of a biosimilar mAb isolated simultaneously |
| Antibody-drug conjugates (ADCs) | DAR 1 – 7 species from DAR distribution | Reference material for individual DAR-species PK/PD and toxicology characterization |
| Peptides | Deletion sequences, oxidized variants, closely eluting process impurities | ICH qualification standards — >50× productivity increase and >50× solvent reduction vs. analytical HPLC |
| Oligonucleotides | n−1 shortmers, n+1 longmers, backbone variants from dsRNA therapeutics | 15× purity increase, 20× mass enrichment |
| Recombinant proteins | PEGylation isoforms, co-eluting process variants | Reference standard preparation at higher purity and concentration than batch methods allow |
| Nutraceuticals / natural products | Low-abundance bioactive compounds in complex matrices | Selective enrichment from natural extracts without requiring affinity steps |
Isolate the Untouchable. Overnight.