









Contichrom® TWIN 1000 LPLC - Polishing
The Contichrom® TWIN 1000 LPLC – Polishing is a sanitary chromatography system for continuous polishing of biologics at production scale — combining the full MCSGP process capabilities of the TWIN HPLC with the sanitary design required for biopharmaceutical manufacturing. MCSGP with AutoPeak® eliminates the yield-purity trade-off, delivering +10–50% absolute yield increase at target purity and up to 70% less buffer consumption compared to batch chromatography. With four LEWA ecodos triplex pumps and a CIP-ready sanitary flow path in SS 316L or Hastelloy, the system is purpose-built for polishing of monoclonal antibodies, viral vectors, ADCs, and recombinant proteins using IEX, HIC, and mixed-mode resins. Available with 7.5-bar maximum operating pressure and 21 CFR Part 11 compliant automation. The TWIN 1000 has flow rates from 0.15 – 18.9 L/min.
- Contichrom® TWIN 300 LPLC - Polishing
- Contichrom® TWIN 500 LPLC - Polishing
- Contichrom® TWIN 1000 LPLC - Polishing
- Contichrom® TWIN 2000 LPLC - Polishing
Overview

Continuous Polishing for Biopharmaceutical Manufacturing
The Contichrom® TWIN LPLC – Polishing is a sanitary chromatography system for continuous polishing of biologics at production scale — combining the full process capabilities of the TWIN HPLC with the sanitary design required for biopharmaceutical manufacturing.
- Twin-column continuous polishing (MCSGP with AutoPeak®)
- Sanitary design with CIP capability, maximum 7.5 bar operating pressure
- 4× LEWA ecodos triplex pumps for full MCSGP operation
- SS 316L or Hastelloy flow paths
- 21 CFR Part 11 compliant software with full audit trail
One System, Multiple Processes
A single sanitary platform for polishing and batch operations:
| Mode | Technology |
|---|---|
| Batch | Single-column batch purification |
| 2D Integrated Batch | Two orthogonal steps, one automated process |
| Continuous Polishing | MCSGP with AutoPeak® |
- Switch between batch, integrated, and MCSGP modes
- AutoPeak® dynamic control ensures consistent quality
- Enables continuous 24/7 manufacturing operation
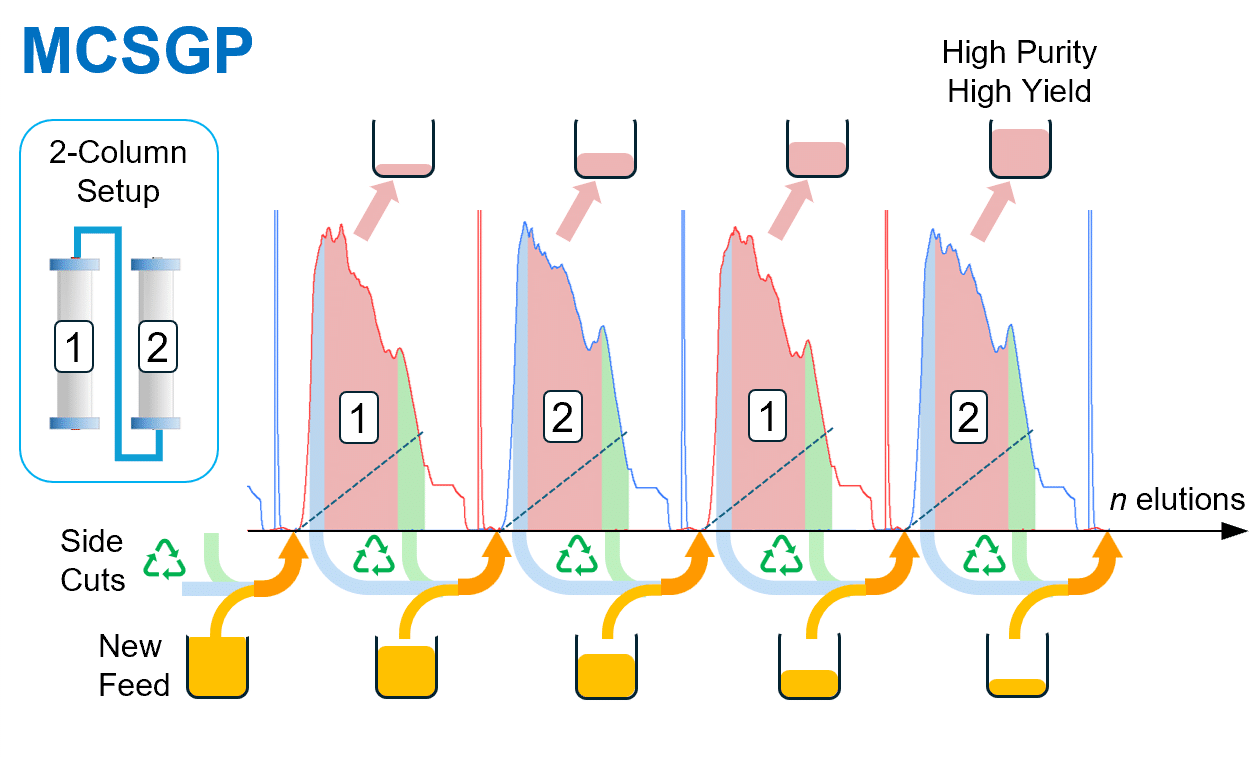
MCSGP: Continuous Polishing at Production Scale
MCSGP with AutoPeak® resolves the purity-yield trade-off:
| Metric | Value |
|---|---|
| Yield | +10–50% absolute yield increase at target purity |
| Solvent/buffer | Up to 70% reduction |
| Re-chromatography | Eliminated |
- Impure side-fractions recycled and re-separated on the second column
- AutoPeak® dynamically adjusts collection windows via real-time UV
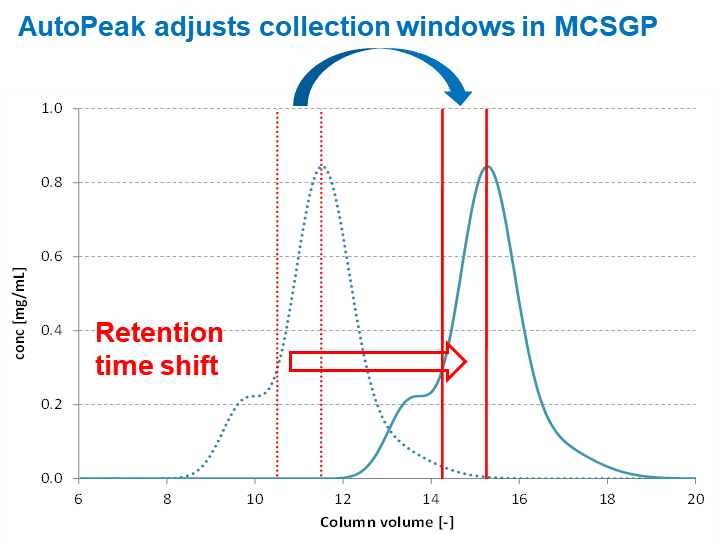
AutoPeak® - Dynamic Process Control
AutoPeak® adjusts collection windows during MCSGP using real-time UV triggers:
- Compensates for process drift, column aging, and feed variability
- Delivers consistent purity and yield without operator intervention
- Minimizes scope for user error during extended production runs
- Enables robust, continuous 24/7 manufacturing
- Makes the process more economical and reliable at commercial scale
Comprehensive Application Range
Production-scale polishing for biopharmaceuticals:
| Application | MCSGP | 2D Integrated Batch |
|---|---|---|
| mAbs & Antibody Variants | ✓ | ✓ |
| Viral Vectors (AAV) | ✓ | ✓ |
| Antibody-drug Conjugates | ✓ | ✓ |
| Recombinant Proteins | ✓ | ✓ |
- All processes run on the same sanitary hardware
- SS 316L or Hastelloy flow paths for chemical compatibility
Scalable from Lab to Production
Part of the Contichrom® platform with established transfer protocols:
| System | Flow Rate | Column ID |
|---|---|---|
| CUBE 30/100 | 0.1–100 mL/min | 0.5–5 cm |
| TWIN LPLC 300 | 0.03-3.33 L/min | 10–20 cm |
| TWIN LPLC 500 | 0.08-8.33 L/min | 20–45 cm |
| TWIN LPLC 1000 | 0.15-18.9 L/min | 30–60 cm |
| TWIN LPLC 2000 | 0.5–36 L/min | 60–100 cm |
- Develop on CUBE, scale up directly to TWIN LPLC Polishing
- Consistent AutoPeak® control principles across all systems
Sanitary Design for Bioprocessing
Purpose-built for biopharmaceutical manufacturing environments:
- Fully sanitary flow path with drainable design and CIP compatibility
- ASME-BPE compliant stainless steel welds on all product contact surfaces
- SS 316L or Hastelloy flow path materials
- SF5 spec product contact tubing (≤ 20 Ra EP)
- Designed for NaOH 1M CIP and compatible with single-use aseptic interfaces
- Maximum operating pressure of 7.5 bar — suitable for IEX, HIC, and mixed-mode resins
Designed for GMP Compliance
Built to meet stringent biopharmaceutical manufacturing requirements:
- Developed under the GAMP 5 framework
- 21 CFR Part 11 compliant software with audit trail
- Comprehensive Turnover Package (TOP) with ASME-BPE compliant welds
- USP Class VI materials and FDA-approved seals
- UL-approved panels, CE marking including PED compliance
TOP includes: MTRs, pressure test reports, passivation reports, weld documentation, as-built drawings.
Production-Grade Hardware
A complete skid-mounted system with four pumps for full MCSGP capability:
- 4× Pumps — LEWA ecodos triplex with rupture detection
- Dual UV Detectors — 4-Channel Adjustable wavelength (200-600 nm), 0.5 mm path length
- 4× Coriolis flow meters — Precision flow monitoring and control
- pH, conductivity & temperature — Post-column standard
- Air sensors — Pre-pump and pre-column
- Pneumatic valves — Sanitary design, fast-acting at low pressure
- 24″ HMI — Industrial touchscreen with FactoryTalk View SE
CIP-Ready Design
The Contichrom® TWIN LPLC – Polishing is designed for cleaning-in-place:
- Dedicated CIP inlet and outlet manifolds integrated into the valve network
- Fully drainable flow path design minimizes carryover between campaigns
- Compatible with NaOH 1M CIP protocols
- Optional single-use aseptic interface for minimally classified suites
- Reduces turnaround time and supports multi-product facility operation
How it works

Sanitary Hardware for GMP Biopharmaceutical Polishing Chromatography
The Contichrom TWIN LPLC – Polishing is a complete sanitary skid-mounted unit for GMP manufacturing:
- Mobile frame with levelers and non-marking caster wheels
- 316L stainless steel frame with mechanically polished welds
- Sanitary flow path designed for drainability and CIP
- Two column positions for MCSGP and independent batch operation
- Operating temperature: 4–25 °C ambient, 4–40 °C process
LEWA Ecodos Triplex Pumps
Four industrial triplex diaphragm pumps for MCSGP and gradient precision:
| Pump | Role |
|---|---|
| Pump A | Gradient component A / Feed |
| Pump B | Gradient component B |
| Pump C | Wash / Regeneration |
| Pump D | Auxiliary / Interconnection flow |
- Triplex configuration for nearly pulse-free flow
- Sandwich diaphragm with rupture detection alarm
- Automatic stroke adjustment for optimal performance
- PTFE diaphragms — USP Class VI certified, FDA approved
- Four pumps enable full MCSGP continuous polishing operation
Pneumatic Valve Network
Sanitary pneumatic valves — fast-acting at low pressure:
- Inlet Selection Valves — 4–6 ports per pump for automated buffer switching
- Column Switching Valves — For MCSGP side-fraction routing and interconnection
- Fraction Outlet Valves — 5+ product/waste outlets with drain
- CIP Manifolds — Dedicated inlet and outlet for cleaning-in-place
- Materials: 316L SS body, PTFE/EPDM diaphragm, SF5 spec (≤ 20 Ra EP)
- At low pressure, standard pneumatic valves provide the rapid actuation MCSGP requires
Dual Wavelength UV Detectors
Two post-column UV sensors for real-time monitoring and AutoPeak® control:
| Parameter | Specification |
|---|---|
| Type | 4-channel, multi-wavelength DAD, 200–600 nm |
| Path length | 0.5 mm path (exchangeable for 0.1, 0.3, or 1 mm) |
- Positioned at the outlet of each column for independent monitoring
- Provides the UV triggers that enable AutoPeak® dynamic process control for MCSGP
In-Line Process Sensors
| Sensor | Quantity | Specification |
|---|---|---|
| Coriolis Flow Meters | 4 | Flow monitoring and control |
| pH | Post-column (×2) | 0–13 pH, ±0.15 unit |
| Conductivity | Post-column (×2) | 0–200 mS/cm, ±1% of measured value |
| Pressure | Multiple | 0–13.8 bar, ±1% FS |
| Temperature | Per column | 0–100 °C, ±0.25 °C |
| Air Sensors | Feed lines | Ultrasonic detection |
- Coriolis meters monitor all four pump flow rates
- pH and conductivity sensors on both outlet channels (waste and collection path) as standard
Column Management
Flexible column configurations for production scale:
- 2 column positions for MCSGP twin-column operation
- Column ID range: 10–100 cm (depending on model)
- 7.5 bar maximum operating pressure
- Sanitary tri-clamp connections for column attachment
- Single-column batch mode available
Human-Machine Interface (HMI)
System control via industrial PC running FactoryTalk View SE:
| Component | Specification |
|---|---|
| Touchscreen | 24″ industrial HMI, panel-mounted |
| Industrial PC | Preloaded with control software |
| OS | Windows 11 Professional |
- Real-time process visualization, recipe management, batch reporting
GMP-Grade Flow Path
Every product-contact surface selected for biopharmaceutical compatibility:
| Component | Materials |
|---|---|
| Product Contact Tubing | 316L SS or Hastelloy, ≤ 20 Ra EP (SF5 spec) |
| Non-Product Contact Tubing | 316L SS, ≤ 20 Ra MP (SF1 spec) |
| Process Valves | 316L SS body, PTFE/EPDM diaphragm |
| Pump Diaphragms | PTFE (USP Class VI, FDA approved) |
- ASME-BPE compliant welds, drainable design, CIP-compatible
- Surface roughness < 0.8 µm Ra on wetted metallic parts
PLC-Based Control System
Industrial automation platform:
| Component | Specification |
|---|---|
| PLC Platform | Rockwell CompactLogix |
| HMI Runtime | FactoryTalk View SE |
| Process Control | MCSGP with AutoPeak® |
| Database | Microsoft SQL Server |
| Historian | YMC Server Historian |
| Reports | YMC Server Reports (auto-generated) |
| Remote Access | HMS Ewon module (client-enabled) |
Optional Hardware
| Option | Specification |
|---|---|
| Buffer In-line Dilution (BID) | Pump E + inlet valves + static mixer + sensors |
| Additional Inlet Valves | Up to 6 per pump |
| Additional Outlet Valves | Up to 5 fraction + 1 waste |
| Filter Housings | Single or dual with valve manifolds |
| Bubble Traps | 2 with valve manifold and vent valves |
| CIP Manifolds | Aseptic single-use bag connection |
| Software Customizations | Handshake with upstream/downstream equipment, additional control loops, external valve/equipment control |
Process Capabilities

Continuous Polishing for Biopharmaceutical Production
The TWIN LPLC – Polishing brings MCSGP continuous polishing to biopharmaceuticals in a sanitary design:
- MCSGP with AutoPeak® — Continuous polishing
- 2D Integrated Batch — Two orthogonal steps, one automated process
- Batch — Single-column gradient and isocratic operation
- All accessible from a single sanitary platform
- Pneumatic valves provide rapid switching at low pressure

MCSGP with AutoPeak® — Continuous Polishing
Twin-column continuous polishing that eliminates the purity-yield trade-off:
- Impure side-fractions automatically identified and recycled to the second column
- Near-complete recovery at target purity — no re-chromatography
- AutoPeak® dynamically adjusts collection windows via real-time UV
- Pneumatic valves switch rapidly at low pressure — ideal for sanitary systems
- Continuous, automated operation for 24/7 manufacturing
Target: Gradient polishing separations (IEX, HIC, mixed-mode) Molecules: mAbs, antibody variants, viral vectors (AAV), ADCs, recombinant proteins
MCSGP — Performance Gains
| Metric | Result |
|---|---|
| Yield | +10–50% absolute yield increase at target purity |
| Buffer consumption | Up to 70% reduction |
| Re-chromatography | Eliminated |
| Column size | Shorter columns — improved productivity |
| Process control | AutoPeak® enables continuous 24/7 operation |
| Quality | Consistent purity regardless of feed variability |
One column loads while the other elutes — continuous output, maximum resin utilization, consistent quality.
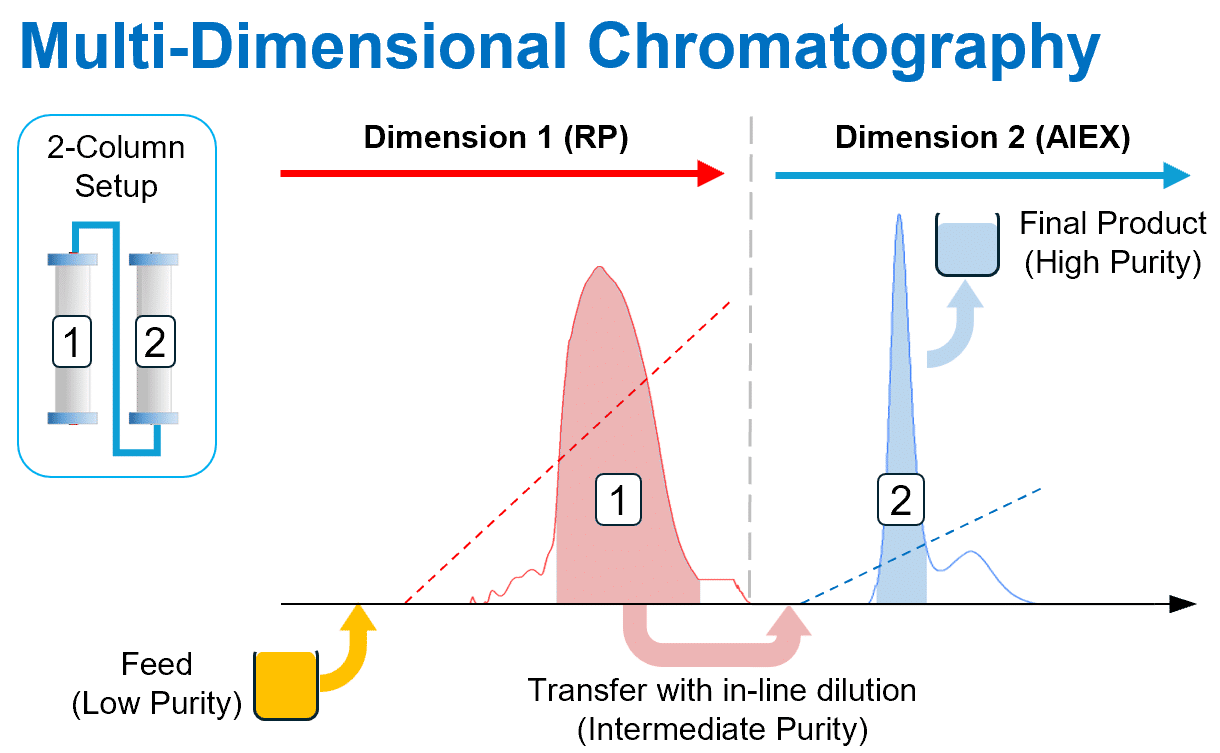
2D Integrated Batch — Multi-Step Automation
Couple two orthogonal chromatography steps in a single automated process:
- Product from column 1 transfers directly to column 2 with in-line dilution
- No intermediate hold tanks or manual handling
- Example: IEX polishing (Dim 1) → HIC polishing (Dim 2)
- Reduces process time, QC burden, and facility footprint
Molecules: mAbs, antibody variants, viral vectors (AAV), ADCs, recombinant proteins
Batch Chromatography
Single-column batch purification:
| Mode | Applications |
|---|---|
| Isocratic | SEC, desalting, affinity |
| Gradient | IEX, HIC, mixed-mode polishing |
- Linear or step gradients
- Multiple fraction collection outlets (5+)
- Full UV/flow/pressure monitoring
Buffer In-line Dilution (BID)
On-board dilution of buffer concentrates and CIP solutions:
- Third pump (Pump E) delivers dilution buffer at controlled ratio
- Static mixer ensures homogeneous blending
- Enables pH adjustment between capture and polish steps
- Supports sequential processing for pool-less multi-step chromatography
Requires: BID option (Pump E, inlet valves, static mixer, sensors)
Regulatory Compliance & Documentation
Certifications
- UL-approved electrical panels
- CE marking including PED compliance
- Developed under GAMP 5 framework
- Complies with FDA 21 CFR Part 11 regulations
Standards & Directives
- ASME-BPE for all stainless steel product contact welds
- FDA-approved USP Class VI materials and seals
- UL-approved electrical panels
- CE marking with PED compliance
Materials & Documentation
- SS 316L or Hastelloy flow paths for biopharmaceutical compatibility
- ASME-BPE compliant welds on all product contact surfaces
- Full traceability with MTRs and certificates of compliance
- Product contact surface: < 0.8 µm Ra (SF5 spec: ≤ 20 Ra EP)
- Turnover Package (TOP) with comprehensive QA/QC documentation
TOP includes: MTRs, certificates of compliance, pressure test reports, passivation reports, weld documentation (logs, inspection reports, isometrics), slope verification, operation and maintenance manuals, as-built drawings (PDF and native CAD/Solidworks).
Specifications
System Models
| Parameter | TWIN 300 | TWIN 500 | TWIN 1000 | TWIN 2000 |
|---|---|---|---|---|
| Flow Rate | 0.03 – 3.33 L/min | 0.08 – 8.33 L/min | 0.15 – 18.9 L/min | 0.5 – 36 L/min |
| Column ID | 10–20 cm | 20–45 cm | 30–60 cm | 60–100 cm |
| Max Operating Pressure | 7.5 bar | 7.5 bar | 7.5 bar | 7.5 bar |
| Best For | Pilot / clinical | Mid-scale GMP | Large-scale GMP | Maximum capacity |
General Specifications
| Specification | Value |
|---|---|
| Max Operating Pressure | 7.5 bar (0.75 MPa / 109 psi) |
| Operating Temperature | 4–25 °C ambient; 4–40 °C process |
| Design | Sanitary, CIP-compatible (NaOH 1M) |
| Wetted Materials | SS 316L or Hastelloy, PTFE, EPDM (USP Class VI) |
| Surface Finish | < 0.8 µm Ra (product contact), SF5 spec: ≤ 20 Ra EP |
| Control System | Rockwell CompactLogix PLC |
| Processes | Batch, 2D Integrated Batch, MCSGP |
Physical Specifications
| Model | Length | Width | Height | Approx. Weight |
|---|---|---|---|---|
| TWIN 300 | 218 cm | 127 cm | 203 cm | 2494 kg |
| TWIN 500 | 299 cm | 165 cm | 198 cm | 3055 kg |
| TWIN 1000 (Pump) | 360 cm | 140 cm | 230 cm | 4455 kg |
| TWIN 1000 (Process) | 140 cm | 90 cm | 200 cm | 1364 kg |
| TWIN 2000 (Pump) | 381 cm | 152 cm | 230 cm | 4763 kg |
| TWIN 2000 (Process) | 147 cm | 101 cm | 200 cm | 1543 kg |
Pumps (LEWA ecodos)
| Configuration | 4× triplex diaphragm pumps |
| Diaphragm | Sandwich PTFE with rupture detection |
| Materials | USP Class VI certified, FDA approved |
| Features | Automatic stroke adjustment, nearly pulse-free flow |
Detectors & Sensors
| UV Detection | 4-channel, multi-wavelength DAD, 200–600 nm, 0.5 mm path (exchangeable for 0.1, 0.3, or 1 mm) |
| Flow Meters (×2) | Coriolis |
| Pressure (×8) | Transmitters throughout flow path, 0–13.8 bar, ±1% FS |
| pH | Post-column, 0–13, ±0.15 unit |
| Conductivity | Post-column, 0–200 mS/cm, ±1% of measured value |
| Temperature | Post-column, 0–100 °C, ±0.25 °C at 25 °C |
| Air Sensors | Pre-pump and pre-column (ultrasonic) |
Valves & Flow Path
| Valve Actuation | Pneumatic diaphragm valves |
| Valve Materials | 316L SS body, PTFE/EPDM diaphragm |
| Product Contact | SF5 spec: ≤ 20 Ra EP |
| Non-Product Contact | SF1 spec: ≤ 20 Ra MP |
| Column Connections | Sanitary tri-clamp |
| CIP | Dedicated inlet and outlet manifolds |
Software & Automation
| PLC Platform | Rockwell CompactLogix |
| HMI Runtime | FactoryTalk View SE |
| Process Control | MCSGP with AutoPeak® |
| Database | Microsoft SQL Server |
| Historian | YMC Server Historian |
| Reports | YMC Server Reports (auto-generated) |
| Remote Access | HMS Ewon module (client-enabled) |
| Data Integrity | 21 CFR Part 11, audit trail, electronic signatures |
User Interface
| HMI | 24″ industrial touchscreen, panel-mounted |
| Industrial PC | Preloaded with control software |
| OS | Windows 11 Professional |
Optional Modules
| Option | Specification |
|---|---|
| Buffer In-line Dilution (BID) | Pump E + inlet valves + static mixer + sensors |
| Additional Inlet Valves | Up to 6 per pump |
| Additional Outlet Valves | Up to 5 fraction + 1 waste |
| Filter Housings | Single or dual with valve manifolds |
| Bubble Traps | 2 with valve manifold and vent valves |
| CIP Manifolds | Aseptic single-use bag connection |
| Software Customizations | Handshake with upstream/downstream equipment, additional control loops, external valve/equipment control |
Technologies

Single-column purification with full method flexibility — gradient, isocratic, step elution. The foundation for process development and the starting point for continuous workflows.
Single-column purification with full method flexibility — gradient, isocratic, step elution. The foundation for process development and the starting point for continuous workflows.

Automate sequential purification across multiple columns and chemistries in a single run. ChromIQ® handles fraction transfer, timing, and method switching.
Automate sequential purification across multiple columns and chemistries in a single run. ChromIQ® handles fraction transfer, timing, and method switching.
Overlapping fractions are automatically recycled between twin columns, breaking the yield–purity tradeoff. AutoPeak® dynamic control maintains performance cycle to cycle.
Overlapping fractions are automatically recycled between twin columns, breaking the yield–purity tradeoff. AutoPeak® dynamic control maintains performance cycle to cycle.

Real-time UV-based control that adapts your continuous process to feed and column variability — eliminating the need for fixed, conservative operating parameters.
Real-time UV-based control that adapts your continuous process to feed and column variability — eliminating the need for fixed, conservative operating parameters.
Applications

MCSGP with AutoPeak® is ideally suited for DAR control, achieving precise drug-antibody ratios with higher yield than batch chromatography.
MCSGP with AutoPeak® is ideally suited for DAR control, achieving precise drug-antibody ratios with higher yield than batch chromatography.

CaptureSMB® with AutomAb® for mAbs & antibody variants: maximize productivity and resin utilization, while slashing buffer consumption.
CaptureSMB® with AutomAb® for mAbs & antibody variants: maximize productivity and resin utilization, while slashing buffer consumption.
From capture through polishing, Contichrom® integrates multiple chromatography steps into automated 2D/3D workflows — reducing hands-on time and intermediate handling.
From capture through polishing, Contichrom® integrates multiple chromatography steps into automated 2D/3D workflows — reducing hands-on time and intermediate handling.

Continuous chromatography improves full/empty capsid separation and recovery, supporting scalable AAV manufacturing.
Continuous chromatography improves full/empty capsid separation and recovery, supporting scalable AAV manufacturing.
Resources
No resources found.
Service & Support

See Contichrom® in action. On-site, at our facility, or remote — get a first-hand look at what our chromatography systems and software can do.
See Contichrom® in action. On-site, at our facility, or remote — get a first-hand look at what our chromatography systems and software can do.

Hands-on operator training, advanced application workshops, and expert consulting — customised to your team's experience and your specific Contichrom® applications.
Hands-on operator training, advanced application workshops, and expert consulting — customised to your team's experience and your specific Contichrom® applications.

Calibrated mechanistic models built from your batch data — simulate and optimize batch, MCSGP, or CaptureSMB® processes computationally, before running a single experiment.
Calibrated mechanistic models built from your batch data — simulate and optimize batch, MCSGP, or CaptureSMB® processes computationally, before running a single experiment.

Try a Contichrom® system in your own lab before committing to a purchase. Full technical support and training included — evaluate the technology on your terms.
Try a Contichrom® system in your own lab before committing to a purchase. Full technical support and training included — evaluate the technology on your terms.

Quantified performance comparison. Run your molecule on our Contichrom® systems and benchmark CaptureSMB® or MCSGP against your existing batch process.
Quantified performance comparison. Run your molecule on our Contichrom® systems and benchmark CaptureSMB® or MCSGP against your existing batch process.

Preventive maintenance subscriptions and on-site repair services to keep your Contichrom® running at peak performance. Remote technical support available worldwide.
Preventive maintenance subscriptions and on-site repair services to keep your Contichrom® running at peak performance. Remote technical support available worldwide.
FAQ
A complete Contichrom® TWIN LPLC Polishing system includes:
- Complete sanitary skid assembly (single or split design depending on model)
- 4× LEWA ecodos triplex pumps with sandwich diaphragms and rupture detection
- Full inlet, column switching, and outlet pneumatic valve network (sanitary design)
- Dual wavelength UV detectors + 4× Coriolis flow meters
- Pressure, temperature, and air sensors
- 24″ HMI with industrial PC and control software preloaded
- Rockwell CompactLogix PLC control system
- CIP inlet and outlet manifolds
- Comprehensive TOP documentation package
- Factory Acceptance Testing (FAT) — up to 5 days
- On-site startup support and training
Customer provides: Columns, filter elements, chromatography resin, external piping, utilities connections.
| Feature | TWIN HPLC | TWIN LPLC Capture | TWIN LPLC Polishing |
|---|---|---|---|
| Design | Industrial (non-sanitary) | Sanitary | Sanitary |
| Pressure | 80 bar | 7.5 bar (20 bar opt.) | 7.5 bar (20 bar opt.) |
| Core Process | MCSGP (polishing) | CaptureSMB (capture) | MCSGP (polishing) |
| HP Valves | YMC fast-acting | Standard pneumatic | Standard pneumatic |
| Pumps | 4× LEWA ecoflow | 2× (3×) LEWA ecodos | 4× LEWA ecodos |
| Molecules | Synthetic (peptides, oligos) | Biologics (mAbs, AAV, ADCs) | Biologics (mAbs, AAV, ADCs) |
| CIP | No | Yes | Yes |
The TWIN LPLC Polishing is the right choice for polishing of biopharmaceuticals where MCSGP continuous polishing, sanitary design, and CIP capability are all required.
| Model | Column ID | Flow Rate | Best For |
|---|---|---|---|
| TWIN 300 | 10–20 cm | 0.03 – 3.33 L/min | Pilot production, clinical manufacturing |
| TWIN 500 | 20–45 cm | 0.15 – 8.33 L/min | Mid-scale commercial production |
| TWIN 1000 | 30–60 cm | 0.15 – 18.9 L/min | Large-scale commercial production |
| TWIN 2000 | 60–100 cm | 0.5 – 36 L/min | Maximum capacity production |
Contact YMC to discuss your throughput requirements and optimal system selection.
- Floor space: Dimensions vary by model (contact YMC for GA drawings)
- Power: Model-dependent (contact YMC for specifications)
- Utilities: Process air, water, compressed air, drain, network connection
- Ambient temperature: 4–25 °C
- Process temperature: 4–40 °C
Note: System must be uncrated, leveled, and connected to utilities before YMC on-site work begins.
- Methods developed on Contichrom CUBE transfer directly to the TWIN LPLC
- Same MCSGP principles and AutoPeak® control at both scales
- YMC ChromaCon provides process transfer support and scale-up assistance
- FAT — Up to 5 days at YMC facility
- On-site startup — One week including commissioning and system walkdown
- Training — 1 full day or 2 half-days covering operation and maintenance
- Remote support — HMS Ewon remote access for troubleshooting and training
- Warranty: 12 months from installation or 18 months from shipment (whichever first)
- Global service: YMC operations worldwide, LEWA support in 80 countries
- Remote access: Built-in capability for automation troubleshooting
- Components: Non-proprietary with global distribution for local acquisition
- Maintenance: No special tools required for standard maintenance
Yes — customization options include:
- Additional inlet valves (up to 6 on Pump A, B, or C)
- Additional outlet/fraction valves (up to 5 fraction + 1 waste)
- Pre-column analytics (pH and conductivity)
- Single or dual filter housings
- Bubble traps
- CIP manifolds for aseptic single-use bag connection
Contact YMC for custom configuration pricing.
Overview

Continuous Polishing for Biopharmaceutical Manufacturing
The Contichrom® TWIN LPLC – Polishing is a sanitary chromatography system for continuous polishing of biologics at production scale — combining the full process capabilities of the TWIN HPLC with the sanitary design required for biopharmaceutical manufacturing.
- Twin-column continuous polishing (MCSGP with AutoPeak®)
- Sanitary design with CIP capability, maximum 7.5 bar operating pressure
- 4× LEWA ecodos triplex pumps for full MCSGP operation
- SS 316L or Hastelloy flow paths
- 21 CFR Part 11 compliant software with full audit trail

One System, Multiple Processes
A single sanitary platform for polishing and batch operations:
| Mode | Technology |
|---|---|
| Batch | Single-column batch purification |
| 2D Integrated Batch | Two orthogonal steps, one automated process |
| Continuous Polishing | MCSGP with AutoPeak® |
- Switch between batch, integrated, and MCSGP modes
- AutoPeak® dynamic control ensures consistent quality
- Enables continuous 24/7 manufacturing operation

MCSGP: Continuous Polishing at Production Scale
MCSGP with AutoPeak® resolves the purity-yield trade-off:
| Metric | Value |
|---|---|
| Yield | +10–50% absolute yield increase at target purity |
| Solvent/buffer | Up to 70% reduction |
| Re-chromatography | Eliminated |
- Impure side-fractions recycled and re-separated on the second column
- AutoPeak® dynamically adjusts collection windows via real-time UV

AutoPeak® - Dynamic Process Control
AutoPeak® adjusts collection windows during MCSGP using real-time UV triggers:
- Compensates for process drift, column aging, and feed variability
- Delivers consistent purity and yield without operator intervention
- Minimizes scope for user error during extended production runs
- Enables robust, continuous 24/7 manufacturing
- Makes the process more economical and reliable at commercial scale
Comprehensive Application Range
Production-scale polishing for biopharmaceuticals:
| Application | MCSGP | 2D Integrated Batch |
|---|---|---|
| mAbs & Antibody Variants | ✓ | ✓ |
| Viral Vectors (AAV) | ✓ | ✓ |
| Antibody-drug Conjugates | ✓ | ✓ |
| Recombinant Proteins | ✓ | ✓ |
- All processes run on the same sanitary hardware
- SS 316L or Hastelloy flow paths for chemical compatibility
Scalable from Lab to Production
Part of the Contichrom® platform with established transfer protocols:
| System | Flow Rate | Column ID |
|---|---|---|
| CUBE 30/100 | 0.1–100 mL/min | 0.5–5 cm |
| TWIN LPLC 300 | 0.03-3.33 L/min | 10–20 cm |
| TWIN LPLC 500 | 0.08-8.33 L/min | 20–45 cm |
| TWIN LPLC 1000 | 0.15-18.9 L/min | 30–60 cm |
| TWIN LPLC 2000 | 0.5–36 L/min | 60–100 cm |
- Develop on CUBE, scale up directly to TWIN LPLC Polishing
- Consistent AutoPeak® control principles across all systems
Sanitary Design for Bioprocessing
Purpose-built for biopharmaceutical manufacturing environments:
- Fully sanitary flow path with drainable design and CIP compatibility
- ASME-BPE compliant stainless steel welds on all product contact surfaces
- SS 316L or Hastelloy flow path materials
- SF5 spec product contact tubing (≤ 20 Ra EP)
- Designed for NaOH 1M CIP and compatible with single-use aseptic interfaces
- Maximum operating pressure of 7.5 bar — suitable for IEX, HIC, and mixed-mode resins
Designed for GMP Compliance
Built to meet stringent biopharmaceutical manufacturing requirements:
- Developed under the GAMP 5 framework
- 21 CFR Part 11 compliant software with audit trail
- Comprehensive Turnover Package (TOP) with ASME-BPE compliant welds
- USP Class VI materials and FDA-approved seals
- UL-approved panels, CE marking including PED compliance
TOP includes: MTRs, pressure test reports, passivation reports, weld documentation, as-built drawings.
Production-Grade Hardware
A complete skid-mounted system with four pumps for full MCSGP capability:
- 4× Pumps — LEWA ecodos triplex with rupture detection
- Dual UV Detectors — 4-Channel Adjustable wavelength (200-600 nm), 0.5 mm path length
- 4× Coriolis flow meters — Precision flow monitoring and control
- pH, conductivity & temperature — Post-column standard
- Air sensors — Pre-pump and pre-column
- Pneumatic valves — Sanitary design, fast-acting at low pressure
- 24″ HMI — Industrial touchscreen with FactoryTalk View SE
CIP-Ready Design
The Contichrom® TWIN LPLC – Polishing is designed for cleaning-in-place:
- Dedicated CIP inlet and outlet manifolds integrated into the valve network
- Fully drainable flow path design minimizes carryover between campaigns
- Compatible with NaOH 1M CIP protocols
- Optional single-use aseptic interface for minimally classified suites
- Reduces turnaround time and supports multi-product facility operation
How it works

Sanitary Hardware for GMP Biopharmaceutical Polishing Chromatography
The Contichrom TWIN LPLC – Polishing is a complete sanitary skid-mounted unit for GMP manufacturing:
- Mobile frame with levelers and non-marking caster wheels
- 316L stainless steel frame with mechanically polished welds
- Sanitary flow path designed for drainability and CIP
- Two column positions for MCSGP and independent batch operation
- Operating temperature: 4–25 °C ambient, 4–40 °C process
LEWA Ecodos Triplex Pumps
Four industrial triplex diaphragm pumps for MCSGP and gradient precision:
| Pump | Role |
|---|---|
| Pump A | Gradient component A / Feed |
| Pump B | Gradient component B |
| Pump C | Wash / Regeneration |
| Pump D | Auxiliary / Interconnection flow |
- Triplex configuration for nearly pulse-free flow
- Sandwich diaphragm with rupture detection alarm
- Automatic stroke adjustment for optimal performance
- PTFE diaphragms — USP Class VI certified, FDA approved
- Four pumps enable full MCSGP continuous polishing operation
Pneumatic Valve Network
Sanitary pneumatic valves — fast-acting at low pressure:
- Inlet Selection Valves — 4–6 ports per pump for automated buffer switching
- Column Switching Valves — For MCSGP side-fraction routing and interconnection
- Fraction Outlet Valves — 5+ product/waste outlets with drain
- CIP Manifolds — Dedicated inlet and outlet for cleaning-in-place
- Materials: 316L SS body, PTFE/EPDM diaphragm, SF5 spec (≤ 20 Ra EP)
- At low pressure, standard pneumatic valves provide the rapid actuation MCSGP requires
Dual Wavelength UV Detectors
Two post-column UV sensors for real-time monitoring and AutoPeak® control:
| Parameter | Specification |
|---|---|
| Type | 4-channel, multi-wavelength DAD, 200–600 nm |
| Path length | 0.5 mm path (exchangeable for 0.1, 0.3, or 1 mm) |
- Positioned at the outlet of each column for independent monitoring
- Provides the UV triggers that enable AutoPeak® dynamic process control for MCSGP
In-Line Process Sensors
| Sensor | Quantity | Specification |
|---|---|---|
| Coriolis Flow Meters | 4 | Flow monitoring and control |
| pH | Post-column (×2) | 0–13 pH, ±0.15 unit |
| Conductivity | Post-column (×2) | 0–200 mS/cm, ±1% of measured value |
| Pressure | Multiple | 0–13.8 bar, ±1% FS |
| Temperature | Per column | 0–100 °C, ±0.25 °C |
| Air Sensors | Feed lines | Ultrasonic detection |
- Coriolis meters monitor all four pump flow rates
- pH and conductivity sensors on both outlet channels (waste and collection path) as standard
Column Management
Flexible column configurations for production scale:
- 2 column positions for MCSGP twin-column operation
- Column ID range: 10–100 cm (depending on model)
- 7.5 bar maximum operating pressure
- Sanitary tri-clamp connections for column attachment
- Single-column batch mode available
Human-Machine Interface (HMI)
System control via industrial PC running FactoryTalk View SE:
| Component | Specification |
|---|---|
| Touchscreen | 24″ industrial HMI, panel-mounted |
| Industrial PC | Preloaded with control software |
| OS | Windows 11 Professional |
- Real-time process visualization, recipe management, batch reporting
GMP-Grade Flow Path
Every product-contact surface selected for biopharmaceutical compatibility:
| Component | Materials |
|---|---|
| Product Contact Tubing | 316L SS or Hastelloy, ≤ 20 Ra EP (SF5 spec) |
| Non-Product Contact Tubing | 316L SS, ≤ 20 Ra MP (SF1 spec) |
| Process Valves | 316L SS body, PTFE/EPDM diaphragm |
| Pump Diaphragms | PTFE (USP Class VI, FDA approved) |
- ASME-BPE compliant welds, drainable design, CIP-compatible
- Surface roughness < 0.8 µm Ra on wetted metallic parts
PLC-Based Control System
Industrial automation platform:
| Component | Specification |
|---|---|
| PLC Platform | Rockwell CompactLogix |
| HMI Runtime | FactoryTalk View SE |
| Process Control | MCSGP with AutoPeak® |
| Database | Microsoft SQL Server |
| Historian | YMC Server Historian |
| Reports | YMC Server Reports (auto-generated) |
| Remote Access | HMS Ewon module (client-enabled) |
Optional Hardware
| Option | Specification |
|---|---|
| Buffer In-line Dilution (BID) | Pump E + inlet valves + static mixer + sensors |
| Additional Inlet Valves | Up to 6 per pump |
| Additional Outlet Valves | Up to 5 fraction + 1 waste |
| Filter Housings | Single or dual with valve manifolds |
| Bubble Traps | 2 with valve manifold and vent valves |
| CIP Manifolds | Aseptic single-use bag connection |
| Software Customizations | Handshake with upstream/downstream equipment, additional control loops, external valve/equipment control |
Process Capabilities

Continuous Polishing for Biopharmaceutical Production
The TWIN LPLC – Polishing brings MCSGP continuous polishing to biopharmaceuticals in a sanitary design:
- MCSGP with AutoPeak® — Continuous polishing
- 2D Integrated Batch — Two orthogonal steps, one automated process
- Batch — Single-column gradient and isocratic operation
- All accessible from a single sanitary platform
- Pneumatic valves provide rapid switching at low pressure

MCSGP with AutoPeak® — Continuous Polishing
Twin-column continuous polishing that eliminates the purity-yield trade-off:
- Impure side-fractions automatically identified and recycled to the second column
- Near-complete recovery at target purity — no re-chromatography
- AutoPeak® dynamically adjusts collection windows via real-time UV
- Pneumatic valves switch rapidly at low pressure — ideal for sanitary systems
- Continuous, automated operation for 24/7 manufacturing
Target: Gradient polishing separations (IEX, HIC, mixed-mode) Molecules: mAbs, antibody variants, viral vectors (AAV), ADCs, recombinant proteins
MCSGP — Performance Gains
| Metric | Result |
|---|---|
| Yield | +10–50% absolute yield increase at target purity |
| Buffer consumption | Up to 70% reduction |
| Re-chromatography | Eliminated |
| Column size | Shorter columns — improved productivity |
| Process control | AutoPeak® enables continuous 24/7 operation |
| Quality | Consistent purity regardless of feed variability |
One column loads while the other elutes — continuous output, maximum resin utilization, consistent quality.

2D Integrated Batch — Multi-Step Automation
Couple two orthogonal chromatography steps in a single automated process:
- Product from column 1 transfers directly to column 2 with in-line dilution
- No intermediate hold tanks or manual handling
- Example: IEX polishing (Dim 1) → HIC polishing (Dim 2)
- Reduces process time, QC burden, and facility footprint
Molecules: mAbs, antibody variants, viral vectors (AAV), ADCs, recombinant proteins
Batch Chromatography
Single-column batch purification:
| Mode | Applications |
|---|---|
| Isocratic | SEC, desalting, affinity |
| Gradient | IEX, HIC, mixed-mode polishing |
- Linear or step gradients
- Multiple fraction collection outlets (5+)
- Full UV/flow/pressure monitoring
Buffer In-line Dilution (BID)
On-board dilution of buffer concentrates and CIP solutions:
- Third pump (Pump E) delivers dilution buffer at controlled ratio
- Static mixer ensures homogeneous blending
- Enables pH adjustment between capture and polish steps
- Supports sequential processing for pool-less multi-step chromatography
Requires: BID option (Pump E, inlet valves, static mixer, sensors)
Regulatory Compliance & Documentation
Certifications
- UL-approved electrical panels
- CE marking including PED compliance
- Developed under GAMP 5 framework
- Complies with FDA 21 CFR Part 11 regulations
Standards & Directives
- ASME-BPE for all stainless steel product contact welds
- FDA-approved USP Class VI materials and seals
- UL-approved electrical panels
- CE marking with PED compliance
Materials & Documentation
- SS 316L or Hastelloy flow paths for biopharmaceutical compatibility
- ASME-BPE compliant welds on all product contact surfaces
- Full traceability with MTRs and certificates of compliance
- Product contact surface: < 0.8 µm Ra (SF5 spec: ≤ 20 Ra EP)
- Turnover Package (TOP) with comprehensive QA/QC documentation
TOP includes: MTRs, certificates of compliance, pressure test reports, passivation reports, weld documentation (logs, inspection reports, isometrics), slope verification, operation and maintenance manuals, as-built drawings (PDF and native CAD/Solidworks).
Specifications
System Models
| Parameter | TWIN 300 | TWIN 500 | TWIN 1000 | TWIN 2000 |
|---|---|---|---|---|
| Flow Rate | 0.03 – 3.33 L/min | 0.08 – 8.33 L/min | 0.15 – 18.9 L/min | 0.5 – 36 L/min |
| Column ID | 10–20 cm | 20–45 cm | 30–60 cm | 60–100 cm |
| Max Operating Pressure | 7.5 bar | 7.5 bar | 7.5 bar | 7.5 bar |
| Best For | Pilot / clinical | Mid-scale GMP | Large-scale GMP | Maximum capacity |
General Specifications
| Specification | Value |
|---|---|
| Max Operating Pressure | 7.5 bar (0.75 MPa / 109 psi) |
| Operating Temperature | 4–25 °C ambient; 4–40 °C process |
| Design | Sanitary, CIP-compatible (NaOH 1M) |
| Wetted Materials | SS 316L or Hastelloy, PTFE, EPDM (USP Class VI) |
| Surface Finish | < 0.8 µm Ra (product contact), SF5 spec: ≤ 20 Ra EP |
| Control System | Rockwell CompactLogix PLC |
| Processes | Batch, 2D Integrated Batch, MCSGP |
Physical Specifications
| Model | Length | Width | Height | Approx. Weight |
|---|---|---|---|---|
| TWIN 300 | 218 cm | 127 cm | 203 cm | 2494 kg |
| TWIN 500 | 299 cm | 165 cm | 198 cm | 3055 kg |
| TWIN 1000 (Pump) | 360 cm | 140 cm | 230 cm | 4455 kg |
| TWIN 1000 (Process) | 140 cm | 90 cm | 200 cm | 1364 kg |
| TWIN 2000 (Pump) | 381 cm | 152 cm | 230 cm | 4763 kg |
| TWIN 2000 (Process) | 147 cm | 101 cm | 200 cm | 1543 kg |
Pumps (LEWA ecodos)
| Configuration | 4× triplex diaphragm pumps |
| Diaphragm | Sandwich PTFE with rupture detection |
| Materials | USP Class VI certified, FDA approved |
| Features | Automatic stroke adjustment, nearly pulse-free flow |
Detectors & Sensors
| UV Detection | 4-channel, multi-wavelength DAD, 200–600 nm, 0.5 mm path (exchangeable for 0.1, 0.3, or 1 mm) |
| Flow Meters (×2) | Coriolis |
| Pressure (×8) | Transmitters throughout flow path, 0–13.8 bar, ±1% FS |
| pH | Post-column, 0–13, ±0.15 unit |
| Conductivity | Post-column, 0–200 mS/cm, ±1% of measured value |
| Temperature | Post-column, 0–100 °C, ±0.25 °C at 25 °C |
| Air Sensors | Pre-pump and pre-column (ultrasonic) |
Valves & Flow Path
| Valve Actuation | Pneumatic diaphragm valves |
| Valve Materials | 316L SS body, PTFE/EPDM diaphragm |
| Product Contact | SF5 spec: ≤ 20 Ra EP |
| Non-Product Contact | SF1 spec: ≤ 20 Ra MP |
| Column Connections | Sanitary tri-clamp |
| CIP | Dedicated inlet and outlet manifolds |
Software & Automation
| PLC Platform | Rockwell CompactLogix |
| HMI Runtime | FactoryTalk View SE |
| Process Control | MCSGP with AutoPeak® |
| Database | Microsoft SQL Server |
| Historian | YMC Server Historian |
| Reports | YMC Server Reports (auto-generated) |
| Remote Access | HMS Ewon module (client-enabled) |
| Data Integrity | 21 CFR Part 11, audit trail, electronic signatures |
User Interface
| HMI | 24″ industrial touchscreen, panel-mounted |
| Industrial PC | Preloaded with control software |
| OS | Windows 11 Professional |
Optional Modules
| Option | Specification |
|---|---|
| Buffer In-line Dilution (BID) | Pump E + inlet valves + static mixer + sensors |
| Additional Inlet Valves | Up to 6 per pump |
| Additional Outlet Valves | Up to 5 fraction + 1 waste |
| Filter Housings | Single or dual with valve manifolds |
| Bubble Traps | 2 with valve manifold and vent valves |
| CIP Manifolds | Aseptic single-use bag connection |
| Software Customizations | Handshake with upstream/downstream equipment, additional control loops, external valve/equipment control |
Technologies

Single-column purification with full method flexibility — gradient, isocratic, step elution. The foundation for process development and the starting point for continuous workflows.
Single-column purification with full method flexibility — gradient, isocratic, step elution. The foundation for process development and the starting point for continuous workflows.

Automate sequential purification across multiple columns and chemistries in a single run. ChromIQ® handles fraction transfer, timing, and method switching.
Automate sequential purification across multiple columns and chemistries in a single run. ChromIQ® handles fraction transfer, timing, and method switching.

Overlapping fractions are automatically recycled between twin columns, breaking the yield–purity tradeoff. AutoPeak® dynamic control maintains performance cycle to cycle.
Overlapping fractions are automatically recycled between twin columns, breaking the yield–purity tradeoff. AutoPeak® dynamic control maintains performance cycle to cycle.

Real-time UV-based control that adapts your continuous process to feed and column variability — eliminating the need for fixed, conservative operating parameters.
Real-time UV-based control that adapts your continuous process to feed and column variability — eliminating the need for fixed, conservative operating parameters.
Applications

MCSGP with AutoPeak® is ideally suited for DAR control, achieving precise drug-antibody ratios with higher yield than batch chromatography.
MCSGP with AutoPeak® is ideally suited for DAR control, achieving precise drug-antibody ratios with higher yield than batch chromatography.

CaptureSMB® with AutomAb® for mAbs & antibody variants: maximize productivity and resin utilization, while slashing buffer consumption.
CaptureSMB® with AutomAb® for mAbs & antibody variants: maximize productivity and resin utilization, while slashing buffer consumption.

From capture through polishing, Contichrom® integrates multiple chromatography steps into automated 2D/3D workflows — reducing hands-on time and intermediate handling.
From capture through polishing, Contichrom® integrates multiple chromatography steps into automated 2D/3D workflows — reducing hands-on time and intermediate handling.

Continuous chromatography improves full/empty capsid separation and recovery, supporting scalable AAV manufacturing.
Continuous chromatography improves full/empty capsid separation and recovery, supporting scalable AAV manufacturing.
Resources
No resources found.
Service & Support

See Contichrom® in action. On-site, at our facility, or remote — get a first-hand look at what our chromatography systems and software can do.
See Contichrom® in action. On-site, at our facility, or remote — get a first-hand look at what our chromatography systems and software can do.

Hands-on operator training, advanced application workshops, and expert consulting — customised to your team's experience and your specific Contichrom® applications.
Hands-on operator training, advanced application workshops, and expert consulting — customised to your team's experience and your specific Contichrom® applications.

Calibrated mechanistic models built from your batch data — simulate and optimize batch, MCSGP, or CaptureSMB® processes computationally, before running a single experiment.
Calibrated mechanistic models built from your batch data — simulate and optimize batch, MCSGP, or CaptureSMB® processes computationally, before running a single experiment.

Try a Contichrom® system in your own lab before committing to a purchase. Full technical support and training included — evaluate the technology on your terms.
Try a Contichrom® system in your own lab before committing to a purchase. Full technical support and training included — evaluate the technology on your terms.

Quantified performance comparison. Run your molecule on our Contichrom® systems and benchmark CaptureSMB® or MCSGP against your existing batch process.
Quantified performance comparison. Run your molecule on our Contichrom® systems and benchmark CaptureSMB® or MCSGP against your existing batch process.

Preventive maintenance subscriptions and on-site repair services to keep your Contichrom® running at peak performance. Remote technical support available worldwide.
Preventive maintenance subscriptions and on-site repair services to keep your Contichrom® running at peak performance. Remote technical support available worldwide.
FAQ
A complete Contichrom® TWIN LPLC Polishing system includes:
- Complete sanitary skid assembly (single or split design depending on model)
- 4× LEWA ecodos triplex pumps with sandwich diaphragms and rupture detection
- Full inlet, column switching, and outlet pneumatic valve network (sanitary design)
- Dual wavelength UV detectors + 4× Coriolis flow meters
- Pressure, temperature, and air sensors
- 24″ HMI with industrial PC and control software preloaded
- Rockwell CompactLogix PLC control system
- CIP inlet and outlet manifolds
- Comprehensive TOP documentation package
- Factory Acceptance Testing (FAT) — up to 5 days
- On-site startup support and training
Customer provides: Columns, filter elements, chromatography resin, external piping, utilities connections.
| Feature | TWIN HPLC | TWIN LPLC Capture | TWIN LPLC Polishing |
|---|---|---|---|
| Design | Industrial (non-sanitary) | Sanitary | Sanitary |
| Pressure | 80 bar | 7.5 bar (20 bar opt.) | 7.5 bar (20 bar opt.) |
| Core Process | MCSGP (polishing) | CaptureSMB (capture) | MCSGP (polishing) |
| HP Valves | YMC fast-acting | Standard pneumatic | Standard pneumatic |
| Pumps | 4× LEWA ecoflow | 2× (3×) LEWA ecodos | 4× LEWA ecodos |
| Molecules | Synthetic (peptides, oligos) | Biologics (mAbs, AAV, ADCs) | Biologics (mAbs, AAV, ADCs) |
| CIP | No | Yes | Yes |
The TWIN LPLC Polishing is the right choice for polishing of biopharmaceuticals where MCSGP continuous polishing, sanitary design, and CIP capability are all required.
| Model | Column ID | Flow Rate | Best For |
|---|---|---|---|
| TWIN 300 | 10–20 cm | 0.03 – 3.33 L/min | Pilot production, clinical manufacturing |
| TWIN 500 | 20–45 cm | 0.15 – 8.33 L/min | Mid-scale commercial production |
| TWIN 1000 | 30–60 cm | 0.15 – 18.9 L/min | Large-scale commercial production |
| TWIN 2000 | 60–100 cm | 0.5 – 36 L/min | Maximum capacity production |
Contact YMC to discuss your throughput requirements and optimal system selection.
- Floor space: Dimensions vary by model (contact YMC for GA drawings)
- Power: Model-dependent (contact YMC for specifications)
- Utilities: Process air, water, compressed air, drain, network connection
- Ambient temperature: 4–25 °C
- Process temperature: 4–40 °C
Note: System must be uncrated, leveled, and connected to utilities before YMC on-site work begins.
- Methods developed on Contichrom CUBE transfer directly to the TWIN LPLC
- Same MCSGP principles and AutoPeak® control at both scales
- YMC ChromaCon provides process transfer support and scale-up assistance
- FAT — Up to 5 days at YMC facility
- On-site startup — One week including commissioning and system walkdown
- Training — 1 full day or 2 half-days covering operation and maintenance
- Remote support — HMS Ewon remote access for troubleshooting and training
- Warranty: 12 months from installation or 18 months from shipment (whichever first)
- Global service: YMC operations worldwide, LEWA support in 80 countries
- Remote access: Built-in capability for automation troubleshooting
- Components: Non-proprietary with global distribution for local acquisition
- Maintenance: No special tools required for standard maintenance
Yes — customization options include:
- Additional inlet valves (up to 6 on Pump A, B, or C)
- Additional outlet/fraction valves (up to 5 fraction + 1 waste)
- Pre-column analytics (pH and conductivity)
- Single or dual filter housings
- Bubble traps
- CIP manifolds for aseptic single-use bag connection
Contact YMC for custom configuration pricing.
Overview

Continuous Polishing for Biopharmaceutical Manufacturing
The Contichrom® TWIN LPLC – Polishing is a sanitary chromatography system for continuous polishing of biologics at production scale — combining the full process capabilities of the TWIN HPLC with the sanitary design required for biopharmaceutical manufacturing.
- Twin-column continuous polishing (MCSGP with AutoPeak®)
- Sanitary design with CIP capability, maximum 7.5 bar operating pressure
- 4× LEWA ecodos triplex pumps for full MCSGP operation
- SS 316L or Hastelloy flow paths
- 21 CFR Part 11 compliant software with full audit trail

One System, Multiple Processes
A single sanitary platform for polishing and batch operations:
| Mode | Technology |
|---|---|
| Batch | Single-column batch purification |
| 2D Integrated Batch | Two orthogonal steps, one automated process |
| Continuous Polishing | MCSGP with AutoPeak® |
- Switch between batch, integrated, and MCSGP modes
- AutoPeak® dynamic control ensures consistent quality
- Enables continuous 24/7 manufacturing operation

MCSGP: Continuous Polishing at Production Scale
MCSGP with AutoPeak® resolves the purity-yield trade-off:
| Metric | Value |
|---|---|
| Yield | +10–50% absolute yield increase at target purity |
| Solvent/buffer | Up to 70% reduction |
| Re-chromatography | Eliminated |
- Impure side-fractions recycled and re-separated on the second column
- AutoPeak® dynamically adjusts collection windows via real-time UV

AutoPeak® - Dynamic Process Control
AutoPeak® adjusts collection windows during MCSGP using real-time UV triggers:
- Compensates for process drift, column aging, and feed variability
- Delivers consistent purity and yield without operator intervention
- Minimizes scope for user error during extended production runs
- Enables robust, continuous 24/7 manufacturing
- Makes the process more economical and reliable at commercial scale
Comprehensive Application Range
Production-scale polishing for biopharmaceuticals:
| Application | MCSGP | 2D Integrated Batch |
|---|---|---|
| mAbs & Antibody Variants | ✓ | ✓ |
| Viral Vectors (AAV) | ✓ | ✓ |
| Antibody-drug Conjugates | ✓ | ✓ |
| Recombinant Proteins | ✓ | ✓ |
- All processes run on the same sanitary hardware
- SS 316L or Hastelloy flow paths for chemical compatibility
Scalable from Lab to Production
Part of the Contichrom® platform with established transfer protocols:
| System | Flow Rate | Column ID |
|---|---|---|
| CUBE 30/100 | 0.1–100 mL/min | 0.5–5 cm |
| TWIN LPLC 300 | 0.03-3.33 L/min | 10–20 cm |
| TWIN LPLC 500 | 0.08-8.33 L/min | 20–45 cm |
| TWIN LPLC 1000 | 0.15-18.9 L/min | 30–60 cm |
| TWIN LPLC 2000 | 0.5–36 L/min | 60–100 cm |
- Develop on CUBE, scale up directly to TWIN LPLC Polishing
- Consistent AutoPeak® control principles across all systems
Sanitary Design for Bioprocessing
Purpose-built for biopharmaceutical manufacturing environments:
- Fully sanitary flow path with drainable design and CIP compatibility
- ASME-BPE compliant stainless steel welds on all product contact surfaces
- SS 316L or Hastelloy flow path materials
- SF5 spec product contact tubing (≤ 20 Ra EP)
- Designed for NaOH 1M CIP and compatible with single-use aseptic interfaces
- Maximum operating pressure of 7.5 bar — suitable for IEX, HIC, and mixed-mode resins
Designed for GMP Compliance
Built to meet stringent biopharmaceutical manufacturing requirements:
- Developed under the GAMP 5 framework
- 21 CFR Part 11 compliant software with audit trail
- Comprehensive Turnover Package (TOP) with ASME-BPE compliant welds
- USP Class VI materials and FDA-approved seals
- UL-approved panels, CE marking including PED compliance
TOP includes: MTRs, pressure test reports, passivation reports, weld documentation, as-built drawings.
Production-Grade Hardware
A complete skid-mounted system with four pumps for full MCSGP capability:
- 4× Pumps — LEWA ecodos triplex with rupture detection
- Dual UV Detectors — 4-Channel Adjustable wavelength (200-600 nm), 0.5 mm path length
- 4× Coriolis flow meters — Precision flow monitoring and control
- pH, conductivity & temperature — Post-column standard
- Air sensors — Pre-pump and pre-column
- Pneumatic valves — Sanitary design, fast-acting at low pressure
- 24″ HMI — Industrial touchscreen with FactoryTalk View SE
CIP-Ready Design
The Contichrom® TWIN LPLC – Polishing is designed for cleaning-in-place:
- Dedicated CIP inlet and outlet manifolds integrated into the valve network
- Fully drainable flow path design minimizes carryover between campaigns
- Compatible with NaOH 1M CIP protocols
- Optional single-use aseptic interface for minimally classified suites
- Reduces turnaround time and supports multi-product facility operation
How it works

Sanitary Hardware for GMP Biopharmaceutical Polishing Chromatography
The Contichrom TWIN LPLC – Polishing is a complete sanitary skid-mounted unit for GMP manufacturing:
- Mobile frame with levelers and non-marking caster wheels
- 316L stainless steel frame with mechanically polished welds
- Sanitary flow path designed for drainability and CIP
- Two column positions for MCSGP and independent batch operation
- Operating temperature: 4–25 °C ambient, 4–40 °C process
LEWA Ecodos Triplex Pumps
Four industrial triplex diaphragm pumps for MCSGP and gradient precision:
| Pump | Role |
|---|---|
| Pump A | Gradient component A / Feed |
| Pump B | Gradient component B |
| Pump C | Wash / Regeneration |
| Pump D | Auxiliary / Interconnection flow |
- Triplex configuration for nearly pulse-free flow
- Sandwich diaphragm with rupture detection alarm
- Automatic stroke adjustment for optimal performance
- PTFE diaphragms — USP Class VI certified, FDA approved
- Four pumps enable full MCSGP continuous polishing operation
Pneumatic Valve Network
Sanitary pneumatic valves — fast-acting at low pressure:
- Inlet Selection Valves — 4–6 ports per pump for automated buffer switching
- Column Switching Valves — For MCSGP side-fraction routing and interconnection
- Fraction Outlet Valves — 5+ product/waste outlets with drain
- CIP Manifolds — Dedicated inlet and outlet for cleaning-in-place
- Materials: 316L SS body, PTFE/EPDM diaphragm, SF5 spec (≤ 20 Ra EP)
- At low pressure, standard pneumatic valves provide the rapid actuation MCSGP requires
Dual Wavelength UV Detectors
Two post-column UV sensors for real-time monitoring and AutoPeak® control:
| Parameter | Specification |
|---|---|
| Type | 4-channel, multi-wavelength DAD, 200–600 nm |
| Path length | 0.5 mm path (exchangeable for 0.1, 0.3, or 1 mm) |
- Positioned at the outlet of each column for independent monitoring
- Provides the UV triggers that enable AutoPeak® dynamic process control for MCSGP
In-Line Process Sensors
| Sensor | Quantity | Specification |
|---|---|---|
| Coriolis Flow Meters | 4 | Flow monitoring and control |
| pH | Post-column (×2) | 0–13 pH, ±0.15 unit |
| Conductivity | Post-column (×2) | 0–200 mS/cm, ±1% of measured value |
| Pressure | Multiple | 0–13.8 bar, ±1% FS |
| Temperature | Per column | 0–100 °C, ±0.25 °C |
| Air Sensors | Feed lines | Ultrasonic detection |
- Coriolis meters monitor all four pump flow rates
- pH and conductivity sensors on both outlet channels (waste and collection path) as standard
Column Management
Flexible column configurations for production scale:
- 2 column positions for MCSGP twin-column operation
- Column ID range: 10–100 cm (depending on model)
- 7.5 bar maximum operating pressure
- Sanitary tri-clamp connections for column attachment
- Single-column batch mode available
Human-Machine Interface (HMI)
System control via industrial PC running FactoryTalk View SE:
| Component | Specification |
|---|---|
| Touchscreen | 24″ industrial HMI, panel-mounted |
| Industrial PC | Preloaded with control software |
| OS | Windows 11 Professional |
- Real-time process visualization, recipe management, batch reporting
GMP-Grade Flow Path
Every product-contact surface selected for biopharmaceutical compatibility:
| Component | Materials |
|---|---|
| Product Contact Tubing | 316L SS or Hastelloy, ≤ 20 Ra EP (SF5 spec) |
| Non-Product Contact Tubing | 316L SS, ≤ 20 Ra MP (SF1 spec) |
| Process Valves | 316L SS body, PTFE/EPDM diaphragm |
| Pump Diaphragms | PTFE (USP Class VI, FDA approved) |
- ASME-BPE compliant welds, drainable design, CIP-compatible
- Surface roughness < 0.8 µm Ra on wetted metallic parts
PLC-Based Control System
Industrial automation platform:
| Component | Specification |
|---|---|
| PLC Platform | Rockwell CompactLogix |
| HMI Runtime | FactoryTalk View SE |
| Process Control | MCSGP with AutoPeak® |
| Database | Microsoft SQL Server |
| Historian | YMC Server Historian |
| Reports | YMC Server Reports (auto-generated) |
| Remote Access | HMS Ewon module (client-enabled) |
Optional Hardware
| Option | Specification |
|---|---|
| Buffer In-line Dilution (BID) | Pump E + inlet valves + static mixer + sensors |
| Additional Inlet Valves | Up to 6 per pump |
| Additional Outlet Valves | Up to 5 fraction + 1 waste |
| Filter Housings | Single or dual with valve manifolds |
| Bubble Traps | 2 with valve manifold and vent valves |
| CIP Manifolds | Aseptic single-use bag connection |
| Software Customizations | Handshake with upstream/downstream equipment, additional control loops, external valve/equipment control |
Process Capabilities

Continuous Polishing for Biopharmaceutical Production
The TWIN LPLC – Polishing brings MCSGP continuous polishing to biopharmaceuticals in a sanitary design:
- MCSGP with AutoPeak® — Continuous polishing
- 2D Integrated Batch — Two orthogonal steps, one automated process
- Batch — Single-column gradient and isocratic operation
- All accessible from a single sanitary platform
- Pneumatic valves provide rapid switching at low pressure

MCSGP with AutoPeak® — Continuous Polishing
Twin-column continuous polishing that eliminates the purity-yield trade-off:
- Impure side-fractions automatically identified and recycled to the second column
- Near-complete recovery at target purity — no re-chromatography
- AutoPeak® dynamically adjusts collection windows via real-time UV
- Pneumatic valves switch rapidly at low pressure — ideal for sanitary systems
- Continuous, automated operation for 24/7 manufacturing
Target: Gradient polishing separations (IEX, HIC, mixed-mode) Molecules: mAbs, antibody variants, viral vectors (AAV), ADCs, recombinant proteins
MCSGP — Performance Gains
| Metric | Result |
|---|---|
| Yield | +10–50% absolute yield increase at target purity |
| Buffer consumption | Up to 70% reduction |
| Re-chromatography | Eliminated |
| Column size | Shorter columns — improved productivity |
| Process control | AutoPeak® enables continuous 24/7 operation |
| Quality | Consistent purity regardless of feed variability |
One column loads while the other elutes — continuous output, maximum resin utilization, consistent quality.

2D Integrated Batch — Multi-Step Automation
Couple two orthogonal chromatography steps in a single automated process:
- Product from column 1 transfers directly to column 2 with in-line dilution
- No intermediate hold tanks or manual handling
- Example: IEX polishing (Dim 1) → HIC polishing (Dim 2)
- Reduces process time, QC burden, and facility footprint
Molecules: mAbs, antibody variants, viral vectors (AAV), ADCs, recombinant proteins
Batch Chromatography
Single-column batch purification:
| Mode | Applications |
|---|---|
| Isocratic | SEC, desalting, affinity |
| Gradient | IEX, HIC, mixed-mode polishing |
- Linear or step gradients
- Multiple fraction collection outlets (5+)
- Full UV/flow/pressure monitoring
Buffer In-line Dilution (BID)
On-board dilution of buffer concentrates and CIP solutions:
- Third pump (Pump E) delivers dilution buffer at controlled ratio
- Static mixer ensures homogeneous blending
- Enables pH adjustment between capture and polish steps
- Supports sequential processing for pool-less multi-step chromatography
Requires: BID option (Pump E, inlet valves, static mixer, sensors)
Regulatory Compliance & Documentation
Certifications
- UL-approved electrical panels
- CE marking including PED compliance
- Developed under GAMP 5 framework
- Complies with FDA 21 CFR Part 11 regulations
Standards & Directives
- ASME-BPE for all stainless steel product contact welds
- FDA-approved USP Class VI materials and seals
- UL-approved electrical panels
- CE marking with PED compliance
Materials & Documentation
- SS 316L or Hastelloy flow paths for biopharmaceutical compatibility
- ASME-BPE compliant welds on all product contact surfaces
- Full traceability with MTRs and certificates of compliance
- Product contact surface: < 0.8 µm Ra (SF5 spec: ≤ 20 Ra EP)
- Turnover Package (TOP) with comprehensive QA/QC documentation
TOP includes: MTRs, certificates of compliance, pressure test reports, passivation reports, weld documentation (logs, inspection reports, isometrics), slope verification, operation and maintenance manuals, as-built drawings (PDF and native CAD/Solidworks).
Specifications
System Models
| Parameter | TWIN 300 | TWIN 500 | TWIN 1000 | TWIN 2000 |
|---|---|---|---|---|
| Flow Rate | 0.03 – 3.33 L/min | 0.08 – 8.33 L/min | 0.15 – 18.9 L/min | 0.5 – 36 L/min |
| Column ID | 10–20 cm | 20–45 cm | 30–60 cm | 60–100 cm |
| Max Operating Pressure | 7.5 bar | 7.5 bar | 7.5 bar | 7.5 bar |
| Best For | Pilot / clinical | Mid-scale GMP | Large-scale GMP | Maximum capacity |
General Specifications
| Specification | Value |
|---|---|
| Max Operating Pressure | 7.5 bar (0.75 MPa / 109 psi) |
| Operating Temperature | 4–25 °C ambient; 4–40 °C process |
| Design | Sanitary, CIP-compatible (NaOH 1M) |
| Wetted Materials | SS 316L or Hastelloy, PTFE, EPDM (USP Class VI) |
| Surface Finish | < 0.8 µm Ra (product contact), SF5 spec: ≤ 20 Ra EP |
| Control System | Rockwell CompactLogix PLC |
| Processes | Batch, 2D Integrated Batch, MCSGP |
Physical Specifications
| Model | Length | Width | Height | Approx. Weight |
|---|---|---|---|---|
| TWIN 300 | 218 cm | 127 cm | 203 cm | 2494 kg |
| TWIN 500 | 299 cm | 165 cm | 198 cm | 3055 kg |
| TWIN 1000 (Pump) | 360 cm | 140 cm | 230 cm | 4455 kg |
| TWIN 1000 (Process) | 140 cm | 90 cm | 200 cm | 1364 kg |
| TWIN 2000 (Pump) | 381 cm | 152 cm | 230 cm | 4763 kg |
| TWIN 2000 (Process) | 147 cm | 101 cm | 200 cm | 1543 kg |
Pumps (LEWA ecodos)
| Configuration | 4× triplex diaphragm pumps |
| Diaphragm | Sandwich PTFE with rupture detection |
| Materials | USP Class VI certified, FDA approved |
| Features | Automatic stroke adjustment, nearly pulse-free flow |
Detectors & Sensors
| UV Detection | 4-channel, multi-wavelength DAD, 200–600 nm, 0.5 mm path (exchangeable for 0.1, 0.3, or 1 mm) |
| Flow Meters (×2) | Coriolis |
| Pressure (×8) | Transmitters throughout flow path, 0–13.8 bar, ±1% FS |
| pH | Post-column, 0–13, ±0.15 unit |
| Conductivity | Post-column, 0–200 mS/cm, ±1% of measured value |
| Temperature | Post-column, 0–100 °C, ±0.25 °C at 25 °C |
| Air Sensors | Pre-pump and pre-column (ultrasonic) |
Valves & Flow Path
| Valve Actuation | Pneumatic diaphragm valves |
| Valve Materials | 316L SS body, PTFE/EPDM diaphragm |
| Product Contact | SF5 spec: ≤ 20 Ra EP |
| Non-Product Contact | SF1 spec: ≤ 20 Ra MP |
| Column Connections | Sanitary tri-clamp |
| CIP | Dedicated inlet and outlet manifolds |
Software & Automation
| PLC Platform | Rockwell CompactLogix |
| HMI Runtime | FactoryTalk View SE |
| Process Control | MCSGP with AutoPeak® |
| Database | Microsoft SQL Server |
| Historian | YMC Server Historian |
| Reports | YMC Server Reports (auto-generated) |
| Remote Access | HMS Ewon module (client-enabled) |
| Data Integrity | 21 CFR Part 11, audit trail, electronic signatures |
User Interface
| HMI | 24″ industrial touchscreen, panel-mounted |
| Industrial PC | Preloaded with control software |
| OS | Windows 11 Professional |
Optional Modules
| Option | Specification |
|---|---|
| Buffer In-line Dilution (BID) | Pump E + inlet valves + static mixer + sensors |
| Additional Inlet Valves | Up to 6 per pump |
| Additional Outlet Valves | Up to 5 fraction + 1 waste |
| Filter Housings | Single or dual with valve manifolds |
| Bubble Traps | 2 with valve manifold and vent valves |
| CIP Manifolds | Aseptic single-use bag connection |
| Software Customizations | Handshake with upstream/downstream equipment, additional control loops, external valve/equipment control |
Technologies

Single-column purification with full method flexibility — gradient, isocratic, step elution. The foundation for process development and the starting point for continuous workflows.
Single-column purification with full method flexibility — gradient, isocratic, step elution. The foundation for process development and the starting point for continuous workflows.

Automate sequential purification across multiple columns and chemistries in a single run. ChromIQ® handles fraction transfer, timing, and method switching.
Automate sequential purification across multiple columns and chemistries in a single run. ChromIQ® handles fraction transfer, timing, and method switching.

Overlapping fractions are automatically recycled between twin columns, breaking the yield–purity tradeoff. AutoPeak® dynamic control maintains performance cycle to cycle.
Overlapping fractions are automatically recycled between twin columns, breaking the yield–purity tradeoff. AutoPeak® dynamic control maintains performance cycle to cycle.

Real-time UV-based control that adapts your continuous process to feed and column variability — eliminating the need for fixed, conservative operating parameters.
Real-time UV-based control that adapts your continuous process to feed and column variability — eliminating the need for fixed, conservative operating parameters.
Applications

MCSGP with AutoPeak® is ideally suited for DAR control, achieving precise drug-antibody ratios with higher yield than batch chromatography.
MCSGP with AutoPeak® is ideally suited for DAR control, achieving precise drug-antibody ratios with higher yield than batch chromatography.

CaptureSMB® with AutomAb® for mAbs & antibody variants: maximize productivity and resin utilization, while slashing buffer consumption.
CaptureSMB® with AutomAb® for mAbs & antibody variants: maximize productivity and resin utilization, while slashing buffer consumption.

From capture through polishing, Contichrom® integrates multiple chromatography steps into automated 2D/3D workflows — reducing hands-on time and intermediate handling.
From capture through polishing, Contichrom® integrates multiple chromatography steps into automated 2D/3D workflows — reducing hands-on time and intermediate handling.

Continuous chromatography improves full/empty capsid separation and recovery, supporting scalable AAV manufacturing.
Continuous chromatography improves full/empty capsid separation and recovery, supporting scalable AAV manufacturing.
Resources
No resources found.
Service & Support

See Contichrom® in action. On-site, at our facility, or remote — get a first-hand look at what our chromatography systems and software can do.
See Contichrom® in action. On-site, at our facility, or remote — get a first-hand look at what our chromatography systems and software can do.

Hands-on operator training, advanced application workshops, and expert consulting — customised to your team's experience and your specific Contichrom® applications.
Hands-on operator training, advanced application workshops, and expert consulting — customised to your team's experience and your specific Contichrom® applications.

Calibrated mechanistic models built from your batch data — simulate and optimize batch, MCSGP, or CaptureSMB® processes computationally, before running a single experiment.
Calibrated mechanistic models built from your batch data — simulate and optimize batch, MCSGP, or CaptureSMB® processes computationally, before running a single experiment.

Try a Contichrom® system in your own lab before committing to a purchase. Full technical support and training included — evaluate the technology on your terms.
Try a Contichrom® system in your own lab before committing to a purchase. Full technical support and training included — evaluate the technology on your terms.

Quantified performance comparison. Run your molecule on our Contichrom® systems and benchmark CaptureSMB® or MCSGP against your existing batch process.
Quantified performance comparison. Run your molecule on our Contichrom® systems and benchmark CaptureSMB® or MCSGP against your existing batch process.

Preventive maintenance subscriptions and on-site repair services to keep your Contichrom® running at peak performance. Remote technical support available worldwide.
Preventive maintenance subscriptions and on-site repair services to keep your Contichrom® running at peak performance. Remote technical support available worldwide.
FAQ
A complete Contichrom® TWIN LPLC Polishing system includes:
- Complete sanitary skid assembly (single or split design depending on model)
- 4× LEWA ecodos triplex pumps with sandwich diaphragms and rupture detection
- Full inlet, column switching, and outlet pneumatic valve network (sanitary design)
- Dual wavelength UV detectors + 4× Coriolis flow meters
- Pressure, temperature, and air sensors
- 24″ HMI with industrial PC and control software preloaded
- Rockwell CompactLogix PLC control system
- CIP inlet and outlet manifolds
- Comprehensive TOP documentation package
- Factory Acceptance Testing (FAT) — up to 5 days
- On-site startup support and training
Customer provides: Columns, filter elements, chromatography resin, external piping, utilities connections.
| Feature | TWIN HPLC | TWIN LPLC Capture | TWIN LPLC Polishing |
|---|---|---|---|
| Design | Industrial (non-sanitary) | Sanitary | Sanitary |
| Pressure | 80 bar | 7.5 bar (20 bar opt.) | 7.5 bar (20 bar opt.) |
| Core Process | MCSGP (polishing) | CaptureSMB (capture) | MCSGP (polishing) |
| HP Valves | YMC fast-acting | Standard pneumatic | Standard pneumatic |
| Pumps | 4× LEWA ecoflow | 2× (3×) LEWA ecodos | 4× LEWA ecodos |
| Molecules | Synthetic (peptides, oligos) | Biologics (mAbs, AAV, ADCs) | Biologics (mAbs, AAV, ADCs) |
| CIP | No | Yes | Yes |
The TWIN LPLC Polishing is the right choice for polishing of biopharmaceuticals where MCSGP continuous polishing, sanitary design, and CIP capability are all required.
| Model | Column ID | Flow Rate | Best For |
|---|---|---|---|
| TWIN 300 | 10–20 cm | 0.03 – 3.33 L/min | Pilot production, clinical manufacturing |
| TWIN 500 | 20–45 cm | 0.15 – 8.33 L/min | Mid-scale commercial production |
| TWIN 1000 | 30–60 cm | 0.15 – 18.9 L/min | Large-scale commercial production |
| TWIN 2000 | 60–100 cm | 0.5 – 36 L/min | Maximum capacity production |
Contact YMC to discuss your throughput requirements and optimal system selection.
- Floor space: Dimensions vary by model (contact YMC for GA drawings)
- Power: Model-dependent (contact YMC for specifications)
- Utilities: Process air, water, compressed air, drain, network connection
- Ambient temperature: 4–25 °C
- Process temperature: 4–40 °C
Note: System must be uncrated, leveled, and connected to utilities before YMC on-site work begins.
- Methods developed on Contichrom CUBE transfer directly to the TWIN LPLC
- Same MCSGP principles and AutoPeak® control at both scales
- YMC ChromaCon provides process transfer support and scale-up assistance
- FAT — Up to 5 days at YMC facility
- On-site startup — One week including commissioning and system walkdown
- Training — 1 full day or 2 half-days covering operation and maintenance
- Remote support — HMS Ewon remote access for troubleshooting and training
- Warranty: 12 months from installation or 18 months from shipment (whichever first)
- Global service: YMC operations worldwide, LEWA support in 80 countries
- Remote access: Built-in capability for automation troubleshooting
- Components: Non-proprietary with global distribution for local acquisition
- Maintenance: No special tools required for standard maintenance
Yes — customization options include:
- Additional inlet valves (up to 6 on Pump A, B, or C)
- Additional outlet/fraction valves (up to 5 fraction + 1 waste)
- Pre-column analytics (pH and conductivity)
- Single or dual filter housings
- Bubble traps
- CIP manifolds for aseptic single-use bag connection
Contact YMC for custom configuration pricing.
Overview

Continuous Polishing for Biopharmaceutical Manufacturing
The Contichrom® TWIN LPLC – Polishing is a sanitary chromatography system for continuous polishing of biologics at production scale — combining the full process capabilities of the TWIN HPLC with the sanitary design required for biopharmaceutical manufacturing.
- Twin-column continuous polishing (MCSGP with AutoPeak®)
- Sanitary design with CIP capability, maximum 7.5 bar operating pressure
- 4× LEWA ecodos triplex pumps for full MCSGP operation
- SS 316L or Hastelloy flow paths
- 21 CFR Part 11 compliant software with full audit trail

One System, Multiple Processes
A single sanitary platform for polishing and batch operations:
| Mode | Technology |
|---|---|
| Batch | Single-column batch purification |
| 2D Integrated Batch | Two orthogonal steps, one automated process |
| Continuous Polishing | MCSGP with AutoPeak® |
- Switch between batch, integrated, and MCSGP modes
- AutoPeak® dynamic control ensures consistent quality
- Enables continuous 24/7 manufacturing operation

MCSGP: Continuous Polishing at Production Scale
MCSGP with AutoPeak® resolves the purity-yield trade-off:
| Metric | Value |
|---|---|
| Yield | +10–50% absolute yield increase at target purity |
| Solvent/buffer | Up to 70% reduction |
| Re-chromatography | Eliminated |
- Impure side-fractions recycled and re-separated on the second column
- AutoPeak® dynamically adjusts collection windows via real-time UV

AutoPeak® - Dynamic Process Control
AutoPeak® adjusts collection windows during MCSGP using real-time UV triggers:
- Compensates for process drift, column aging, and feed variability
- Delivers consistent purity and yield without operator intervention
- Minimizes scope for user error during extended production runs
- Enables robust, continuous 24/7 manufacturing
- Makes the process more economical and reliable at commercial scale
Comprehensive Application Range
Production-scale polishing for biopharmaceuticals:
| Application | MCSGP | 2D Integrated Batch |
|---|---|---|
| mAbs & Antibody Variants | ✓ | ✓ |
| Viral Vectors (AAV) | ✓ | ✓ |
| Antibody-drug Conjugates | ✓ | ✓ |
| Recombinant Proteins | ✓ | ✓ |
- All processes run on the same sanitary hardware
- SS 316L or Hastelloy flow paths for chemical compatibility
Scalable from Lab to Production
Part of the Contichrom® platform with established transfer protocols:
| System | Flow Rate | Column ID |
|---|---|---|
| CUBE 30/100 | 0.1–100 mL/min | 0.5–5 cm |
| TWIN LPLC 300 | 0.03-3.33 L/min | 10–20 cm |
| TWIN LPLC 500 | 0.08-8.33 L/min | 20–45 cm |
| TWIN LPLC 1000 | 0.15-18.9 L/min | 30–60 cm |
| TWIN LPLC 2000 | 0.5–36 L/min | 60–100 cm |
- Develop on CUBE, scale up directly to TWIN LPLC Polishing
- Consistent AutoPeak® control principles across all systems
Sanitary Design for Bioprocessing
Purpose-built for biopharmaceutical manufacturing environments:
- Fully sanitary flow path with drainable design and CIP compatibility
- ASME-BPE compliant stainless steel welds on all product contact surfaces
- SS 316L or Hastelloy flow path materials
- SF5 spec product contact tubing (≤ 20 Ra EP)
- Designed for NaOH 1M CIP and compatible with single-use aseptic interfaces
- Maximum operating pressure of 7.5 bar — suitable for IEX, HIC, and mixed-mode resins
Designed for GMP Compliance
Built to meet stringent biopharmaceutical manufacturing requirements:
- Developed under the GAMP 5 framework
- 21 CFR Part 11 compliant software with audit trail
- Comprehensive Turnover Package (TOP) with ASME-BPE compliant welds
- USP Class VI materials and FDA-approved seals
- UL-approved panels, CE marking including PED compliance
TOP includes: MTRs, pressure test reports, passivation reports, weld documentation, as-built drawings.
Production-Grade Hardware
A complete skid-mounted system with four pumps for full MCSGP capability:
- 4× Pumps — LEWA ecodos triplex with rupture detection
- Dual UV Detectors — 4-Channel Adjustable wavelength (200-600 nm), 0.5 mm path length
- 4× Coriolis flow meters — Precision flow monitoring and control
- pH, conductivity & temperature — Post-column standard
- Air sensors — Pre-pump and pre-column
- Pneumatic valves — Sanitary design, fast-acting at low pressure
- 24″ HMI — Industrial touchscreen with FactoryTalk View SE
CIP-Ready Design
The Contichrom® TWIN LPLC – Polishing is designed for cleaning-in-place:
- Dedicated CIP inlet and outlet manifolds integrated into the valve network
- Fully drainable flow path design minimizes carryover between campaigns
- Compatible with NaOH 1M CIP protocols
- Optional single-use aseptic interface for minimally classified suites
- Reduces turnaround time and supports multi-product facility operation
How it works

Sanitary Hardware for GMP Biopharmaceutical Polishing Chromatography
The Contichrom TWIN LPLC – Polishing is a complete sanitary skid-mounted unit for GMP manufacturing:
- Mobile frame with levelers and non-marking caster wheels
- 316L stainless steel frame with mechanically polished welds
- Sanitary flow path designed for drainability and CIP
- Two column positions for MCSGP and independent batch operation
- Operating temperature: 4–25 °C ambient, 4–40 °C process
LEWA Ecodos Triplex Pumps
Four industrial triplex diaphragm pumps for MCSGP and gradient precision:
| Pump | Role |
|---|---|
| Pump A | Gradient component A / Feed |
| Pump B | Gradient component B |
| Pump C | Wash / Regeneration |
| Pump D | Auxiliary / Interconnection flow |
- Triplex configuration for nearly pulse-free flow
- Sandwich diaphragm with rupture detection alarm
- Automatic stroke adjustment for optimal performance
- PTFE diaphragms — USP Class VI certified, FDA approved
- Four pumps enable full MCSGP continuous polishing operation
Pneumatic Valve Network
Sanitary pneumatic valves — fast-acting at low pressure:
- Inlet Selection Valves — 4–6 ports per pump for automated buffer switching
- Column Switching Valves — For MCSGP side-fraction routing and interconnection
- Fraction Outlet Valves — 5+ product/waste outlets with drain
- CIP Manifolds — Dedicated inlet and outlet for cleaning-in-place
- Materials: 316L SS body, PTFE/EPDM diaphragm, SF5 spec (≤ 20 Ra EP)
- At low pressure, standard pneumatic valves provide the rapid actuation MCSGP requires
Dual Wavelength UV Detectors
Two post-column UV sensors for real-time monitoring and AutoPeak® control:
| Parameter | Specification |
|---|---|
| Type | 4-channel, multi-wavelength DAD, 200–600 nm |
| Path length | 0.5 mm path (exchangeable for 0.1, 0.3, or 1 mm) |
- Positioned at the outlet of each column for independent monitoring
- Provides the UV triggers that enable AutoPeak® dynamic process control for MCSGP
In-Line Process Sensors
| Sensor | Quantity | Specification |
|---|---|---|
| Coriolis Flow Meters | 4 | Flow monitoring and control |
| pH | Post-column (×2) | 0–13 pH, ±0.15 unit |
| Conductivity | Post-column (×2) | 0–200 mS/cm, ±1% of measured value |
| Pressure | Multiple | 0–13.8 bar, ±1% FS |
| Temperature | Per column | 0–100 °C, ±0.25 °C |
| Air Sensors | Feed lines | Ultrasonic detection |
- Coriolis meters monitor all four pump flow rates
- pH and conductivity sensors on both outlet channels (waste and collection path) as standard
Column Management
Flexible column configurations for production scale:
- 2 column positions for MCSGP twin-column operation
- Column ID range: 10–100 cm (depending on model)
- 7.5 bar maximum operating pressure
- Sanitary tri-clamp connections for column attachment
- Single-column batch mode available
Human-Machine Interface (HMI)
System control via industrial PC running FactoryTalk View SE:
| Component | Specification |
|---|---|
| Touchscreen | 24″ industrial HMI, panel-mounted |
| Industrial PC | Preloaded with control software |
| OS | Windows 11 Professional |
- Real-time process visualization, recipe management, batch reporting
GMP-Grade Flow Path
Every product-contact surface selected for biopharmaceutical compatibility:
| Component | Materials |
|---|---|
| Product Contact Tubing | 316L SS or Hastelloy, ≤ 20 Ra EP (SF5 spec) |
| Non-Product Contact Tubing | 316L SS, ≤ 20 Ra MP (SF1 spec) |
| Process Valves | 316L SS body, PTFE/EPDM diaphragm |
| Pump Diaphragms | PTFE (USP Class VI, FDA approved) |
- ASME-BPE compliant welds, drainable design, CIP-compatible
- Surface roughness < 0.8 µm Ra on wetted metallic parts
PLC-Based Control System
Industrial automation platform:
| Component | Specification |
|---|---|
| PLC Platform | Rockwell CompactLogix |
| HMI Runtime | FactoryTalk View SE |
| Process Control | MCSGP with AutoPeak® |
| Database | Microsoft SQL Server |
| Historian | YMC Server Historian |
| Reports | YMC Server Reports (auto-generated) |
| Remote Access | HMS Ewon module (client-enabled) |
Optional Hardware
| Option | Specification |
|---|---|
| Buffer In-line Dilution (BID) | Pump E + inlet valves + static mixer + sensors |
| Additional Inlet Valves | Up to 6 per pump |
| Additional Outlet Valves | Up to 5 fraction + 1 waste |
| Filter Housings | Single or dual with valve manifolds |
| Bubble Traps | 2 with valve manifold and vent valves |
| CIP Manifolds | Aseptic single-use bag connection |
| Software Customizations | Handshake with upstream/downstream equipment, additional control loops, external valve/equipment control |
Process Capabilities

Continuous Polishing for Biopharmaceutical Production
The TWIN LPLC – Polishing brings MCSGP continuous polishing to biopharmaceuticals in a sanitary design:
- MCSGP with AutoPeak® — Continuous polishing
- 2D Integrated Batch — Two orthogonal steps, one automated process
- Batch — Single-column gradient and isocratic operation
- All accessible from a single sanitary platform
- Pneumatic valves provide rapid switching at low pressure

MCSGP with AutoPeak® — Continuous Polishing
Twin-column continuous polishing that eliminates the purity-yield trade-off:
- Impure side-fractions automatically identified and recycled to the second column
- Near-complete recovery at target purity — no re-chromatography
- AutoPeak® dynamically adjusts collection windows via real-time UV
- Pneumatic valves switch rapidly at low pressure — ideal for sanitary systems
- Continuous, automated operation for 24/7 manufacturing
Target: Gradient polishing separations (IEX, HIC, mixed-mode) Molecules: mAbs, antibody variants, viral vectors (AAV), ADCs, recombinant proteins
MCSGP — Performance Gains
| Metric | Result |
|---|---|
| Yield | +10–50% absolute yield increase at target purity |
| Buffer consumption | Up to 70% reduction |
| Re-chromatography | Eliminated |
| Column size | Shorter columns — improved productivity |
| Process control | AutoPeak® enables continuous 24/7 operation |
| Quality | Consistent purity regardless of feed variability |
One column loads while the other elutes — continuous output, maximum resin utilization, consistent quality.

2D Integrated Batch — Multi-Step Automation
Couple two orthogonal chromatography steps in a single automated process:
- Product from column 1 transfers directly to column 2 with in-line dilution
- No intermediate hold tanks or manual handling
- Example: IEX polishing (Dim 1) → HIC polishing (Dim 2)
- Reduces process time, QC burden, and facility footprint
Molecules: mAbs, antibody variants, viral vectors (AAV), ADCs, recombinant proteins
Batch Chromatography
Single-column batch purification:
| Mode | Applications |
|---|---|
| Isocratic | SEC, desalting, affinity |
| Gradient | IEX, HIC, mixed-mode polishing |
- Linear or step gradients
- Multiple fraction collection outlets (5+)
- Full UV/flow/pressure monitoring
Buffer In-line Dilution (BID)
On-board dilution of buffer concentrates and CIP solutions:
- Third pump (Pump E) delivers dilution buffer at controlled ratio
- Static mixer ensures homogeneous blending
- Enables pH adjustment between capture and polish steps
- Supports sequential processing for pool-less multi-step chromatography
Requires: BID option (Pump E, inlet valves, static mixer, sensors)
Regulatory Compliance & Documentation
Certifications
- UL-approved electrical panels
- CE marking including PED compliance
- Developed under GAMP 5 framework
- Complies with FDA 21 CFR Part 11 regulations
Standards & Directives
- ASME-BPE for all stainless steel product contact welds
- FDA-approved USP Class VI materials and seals
- UL-approved electrical panels
- CE marking with PED compliance
Materials & Documentation
- SS 316L or Hastelloy flow paths for biopharmaceutical compatibility
- ASME-BPE compliant welds on all product contact surfaces
- Full traceability with MTRs and certificates of compliance
- Product contact surface: < 0.8 µm Ra (SF5 spec: ≤ 20 Ra EP)
- Turnover Package (TOP) with comprehensive QA/QC documentation
TOP includes: MTRs, certificates of compliance, pressure test reports, passivation reports, weld documentation (logs, inspection reports, isometrics), slope verification, operation and maintenance manuals, as-built drawings (PDF and native CAD/Solidworks).
Specifications
System Models
| Parameter | TWIN 300 | TWIN 500 | TWIN 1000 | TWIN 2000 |
|---|---|---|---|---|
| Flow Rate | 0.03 – 3.33 L/min | 0.08 – 8.33 L/min | 0.15 – 18.9 L/min | 0.5 – 36 L/min |
| Column ID | 10–20 cm | 20–45 cm | 30–60 cm | 60–100 cm |
| Max Operating Pressure | 7.5 bar | 7.5 bar | 7.5 bar | 7.5 bar |
| Best For | Pilot / clinical | Mid-scale GMP | Large-scale GMP | Maximum capacity |
General Specifications
| Specification | Value |
|---|---|
| Max Operating Pressure | 7.5 bar (0.75 MPa / 109 psi) |
| Operating Temperature | 4–25 °C ambient; 4–40 °C process |
| Design | Sanitary, CIP-compatible (NaOH 1M) |
| Wetted Materials | SS 316L or Hastelloy, PTFE, EPDM (USP Class VI) |
| Surface Finish | < 0.8 µm Ra (product contact), SF5 spec: ≤ 20 Ra EP |
| Control System | Rockwell CompactLogix PLC |
| Processes | Batch, 2D Integrated Batch, MCSGP |
Physical Specifications
| Model | Length | Width | Height | Approx. Weight |
|---|---|---|---|---|
| TWIN 300 | 218 cm | 127 cm | 203 cm | 2494 kg |
| TWIN 500 | 299 cm | 165 cm | 198 cm | 3055 kg |
| TWIN 1000 (Pump) | 360 cm | 140 cm | 230 cm | 4455 kg |
| TWIN 1000 (Process) | 140 cm | 90 cm | 200 cm | 1364 kg |
| TWIN 2000 (Pump) | 381 cm | 152 cm | 230 cm | 4763 kg |
| TWIN 2000 (Process) | 147 cm | 101 cm | 200 cm | 1543 kg |
Pumps (LEWA ecodos)
| Configuration | 4× triplex diaphragm pumps |
| Diaphragm | Sandwich PTFE with rupture detection |
| Materials | USP Class VI certified, FDA approved |
| Features | Automatic stroke adjustment, nearly pulse-free flow |
Detectors & Sensors
| UV Detection | 4-channel, multi-wavelength DAD, 200–600 nm, 0.5 mm path (exchangeable for 0.1, 0.3, or 1 mm) |
| Flow Meters (×2) | Coriolis |
| Pressure (×8) | Transmitters throughout flow path, 0–13.8 bar, ±1% FS |
| pH | Post-column, 0–13, ±0.15 unit |
| Conductivity | Post-column, 0–200 mS/cm, ±1% of measured value |
| Temperature | Post-column, 0–100 °C, ±0.25 °C at 25 °C |
| Air Sensors | Pre-pump and pre-column (ultrasonic) |
Valves & Flow Path
| Valve Actuation | Pneumatic diaphragm valves |
| Valve Materials | 316L SS body, PTFE/EPDM diaphragm |
| Product Contact | SF5 spec: ≤ 20 Ra EP |
| Non-Product Contact | SF1 spec: ≤ 20 Ra MP |
| Column Connections | Sanitary tri-clamp |
| CIP | Dedicated inlet and outlet manifolds |
Software & Automation
| PLC Platform | Rockwell CompactLogix |
| HMI Runtime | FactoryTalk View SE |
| Process Control | MCSGP with AutoPeak® |
| Database | Microsoft SQL Server |
| Historian | YMC Server Historian |
| Reports | YMC Server Reports (auto-generated) |
| Remote Access | HMS Ewon module (client-enabled) |
| Data Integrity | 21 CFR Part 11, audit trail, electronic signatures |
User Interface
| HMI | 24″ industrial touchscreen, panel-mounted |
| Industrial PC | Preloaded with control software |
| OS | Windows 11 Professional |
Optional Modules
| Option | Specification |
|---|---|
| Buffer In-line Dilution (BID) | Pump E + inlet valves + static mixer + sensors |
| Additional Inlet Valves | Up to 6 per pump |
| Additional Outlet Valves | Up to 5 fraction + 1 waste |
| Filter Housings | Single or dual with valve manifolds |
| Bubble Traps | 2 with valve manifold and vent valves |
| CIP Manifolds | Aseptic single-use bag connection |
| Software Customizations | Handshake with upstream/downstream equipment, additional control loops, external valve/equipment control |
Technologies

Single-column purification with full method flexibility — gradient, isocratic, step elution. The foundation for process development and the starting point for continuous workflows.
Single-column purification with full method flexibility — gradient, isocratic, step elution. The foundation for process development and the starting point for continuous workflows.

Automate sequential purification across multiple columns and chemistries in a single run. ChromIQ® handles fraction transfer, timing, and method switching.
Automate sequential purification across multiple columns and chemistries in a single run. ChromIQ® handles fraction transfer, timing, and method switching.

Overlapping fractions are automatically recycled between twin columns, breaking the yield–purity tradeoff. AutoPeak® dynamic control maintains performance cycle to cycle.
Overlapping fractions are automatically recycled between twin columns, breaking the yield–purity tradeoff. AutoPeak® dynamic control maintains performance cycle to cycle.

Real-time UV-based control that adapts your continuous process to feed and column variability — eliminating the need for fixed, conservative operating parameters.
Real-time UV-based control that adapts your continuous process to feed and column variability — eliminating the need for fixed, conservative operating parameters.
Applications

MCSGP with AutoPeak® is ideally suited for DAR control, achieving precise drug-antibody ratios with higher yield than batch chromatography.
MCSGP with AutoPeak® is ideally suited for DAR control, achieving precise drug-antibody ratios with higher yield than batch chromatography.

CaptureSMB® with AutomAb® for mAbs & antibody variants: maximize productivity and resin utilization, while slashing buffer consumption.
CaptureSMB® with AutomAb® for mAbs & antibody variants: maximize productivity and resin utilization, while slashing buffer consumption.

From capture through polishing, Contichrom® integrates multiple chromatography steps into automated 2D/3D workflows — reducing hands-on time and intermediate handling.
From capture through polishing, Contichrom® integrates multiple chromatography steps into automated 2D/3D workflows — reducing hands-on time and intermediate handling.

Continuous chromatography improves full/empty capsid separation and recovery, supporting scalable AAV manufacturing.
Continuous chromatography improves full/empty capsid separation and recovery, supporting scalable AAV manufacturing.
Resources
No resources found.
Service & Support

See Contichrom® in action. On-site, at our facility, or remote — get a first-hand look at what our chromatography systems and software can do.
See Contichrom® in action. On-site, at our facility, or remote — get a first-hand look at what our chromatography systems and software can do.

Hands-on operator training, advanced application workshops, and expert consulting — customised to your team's experience and your specific Contichrom® applications.
Hands-on operator training, advanced application workshops, and expert consulting — customised to your team's experience and your specific Contichrom® applications.

Calibrated mechanistic models built from your batch data — simulate and optimize batch, MCSGP, or CaptureSMB® processes computationally, before running a single experiment.
Calibrated mechanistic models built from your batch data — simulate and optimize batch, MCSGP, or CaptureSMB® processes computationally, before running a single experiment.

Try a Contichrom® system in your own lab before committing to a purchase. Full technical support and training included — evaluate the technology on your terms.
Try a Contichrom® system in your own lab before committing to a purchase. Full technical support and training included — evaluate the technology on your terms.

Quantified performance comparison. Run your molecule on our Contichrom® systems and benchmark CaptureSMB® or MCSGP against your existing batch process.
Quantified performance comparison. Run your molecule on our Contichrom® systems and benchmark CaptureSMB® or MCSGP against your existing batch process.

Preventive maintenance subscriptions and on-site repair services to keep your Contichrom® running at peak performance. Remote technical support available worldwide.
Preventive maintenance subscriptions and on-site repair services to keep your Contichrom® running at peak performance. Remote technical support available worldwide.
FAQ
A complete Contichrom® TWIN LPLC Polishing system includes:
- Complete sanitary skid assembly (single or split design depending on model)
- 4× LEWA ecodos triplex pumps with sandwich diaphragms and rupture detection
- Full inlet, column switching, and outlet pneumatic valve network (sanitary design)
- Dual wavelength UV detectors + 4× Coriolis flow meters
- Pressure, temperature, and air sensors
- 24″ HMI with industrial PC and control software preloaded
- Rockwell CompactLogix PLC control system
- CIP inlet and outlet manifolds
- Comprehensive TOP documentation package
- Factory Acceptance Testing (FAT) — up to 5 days
- On-site startup support and training
Customer provides: Columns, filter elements, chromatography resin, external piping, utilities connections.
| Feature | TWIN HPLC | TWIN LPLC Capture | TWIN LPLC Polishing |
|---|---|---|---|
| Design | Industrial (non-sanitary) | Sanitary | Sanitary |
| Pressure | 80 bar | 7.5 bar (20 bar opt.) | 7.5 bar (20 bar opt.) |
| Core Process | MCSGP (polishing) | CaptureSMB (capture) | MCSGP (polishing) |
| HP Valves | YMC fast-acting | Standard pneumatic | Standard pneumatic |
| Pumps | 4× LEWA ecoflow | 2× (3×) LEWA ecodos | 4× LEWA ecodos |
| Molecules | Synthetic (peptides, oligos) | Biologics (mAbs, AAV, ADCs) | Biologics (mAbs, AAV, ADCs) |
| CIP | No | Yes | Yes |
The TWIN LPLC Polishing is the right choice for polishing of biopharmaceuticals where MCSGP continuous polishing, sanitary design, and CIP capability are all required.
| Model | Column ID | Flow Rate | Best For |
|---|---|---|---|
| TWIN 300 | 10–20 cm | 0.03 – 3.33 L/min | Pilot production, clinical manufacturing |
| TWIN 500 | 20–45 cm | 0.15 – 8.33 L/min | Mid-scale commercial production |
| TWIN 1000 | 30–60 cm | 0.15 – 18.9 L/min | Large-scale commercial production |
| TWIN 2000 | 60–100 cm | 0.5 – 36 L/min | Maximum capacity production |
Contact YMC to discuss your throughput requirements and optimal system selection.
- Floor space: Dimensions vary by model (contact YMC for GA drawings)
- Power: Model-dependent (contact YMC for specifications)
- Utilities: Process air, water, compressed air, drain, network connection
- Ambient temperature: 4–25 °C
- Process temperature: 4–40 °C
Note: System must be uncrated, leveled, and connected to utilities before YMC on-site work begins.
- Methods developed on Contichrom CUBE transfer directly to the TWIN LPLC
- Same MCSGP principles and AutoPeak® control at both scales
- YMC ChromaCon provides process transfer support and scale-up assistance
- FAT — Up to 5 days at YMC facility
- On-site startup — One week including commissioning and system walkdown
- Training — 1 full day or 2 half-days covering operation and maintenance
- Remote support — HMS Ewon remote access for troubleshooting and training
- Warranty: 12 months from installation or 18 months from shipment (whichever first)
- Global service: YMC operations worldwide, LEWA support in 80 countries
- Remote access: Built-in capability for automation troubleshooting
- Components: Non-proprietary with global distribution for local acquisition
- Maintenance: No special tools required for standard maintenance
Yes — customization options include:
- Additional inlet valves (up to 6 on Pump A, B, or C)
- Additional outlet/fraction valves (up to 5 fraction + 1 waste)
- Pre-column analytics (pH and conductivity)
- Single or dual filter housings
- Bubble traps
- CIP manifolds for aseptic single-use bag connection
Contact YMC for custom configuration pricing.


