









Contichrom® TWIN 500 LPLC - Capture
The Contichrom® TWIN 500 LPLC – Capture is a sanitary chromatography system for continuous capture of biologics from pilot to large-scale GMP manufacturing. Featuring patented CaptureSMB® technology with AutomAb® dynamic control, the system maximizes resin binding capacity to deliver up to 3× faster processing and 50% less Protein A resin and buffer consumption compared to batch chromatography. Purpose-built for monoclonal antibodies, viral vectors (AAV), and recombinant proteins, the system features a fully sanitary, CIP-ready flow path in SS 316L or Hastelloy with a 7.5-bar maximum operating pressure. The TWIN 500 has flow rates from 0.08 – 8.33 L/min.
- Contichrom® TWIN 300 LPLC - Capture
- Contichrom® TWIN 500 LPLC - Capture
- Contichrom® TWIN 1000 LPLC - Capture
- Contichrom® TWIN 2000 LPLC - Capture
Overview

GMP-Ready Continuous Capture for Biopharmaceutical Manufacturing
The Contichrom® TWIN LPLC – Capture is a sanitary chromatography system for continuous capture of biologics from pilot to large-scale GMP manufacturing.
- Twin-column continuous capture (CaptureSMB® with AutomAb®)
- Up to 3× faster processing, 50% less resin and buffer vs. batch
- Sanitary design with CIP capability for biopharmaceutical production
- SS 316L or Hastelloy flow paths, 7.5 bar maximum operating pressure
- 21 CFR Part 11 compliant software with full audit trail
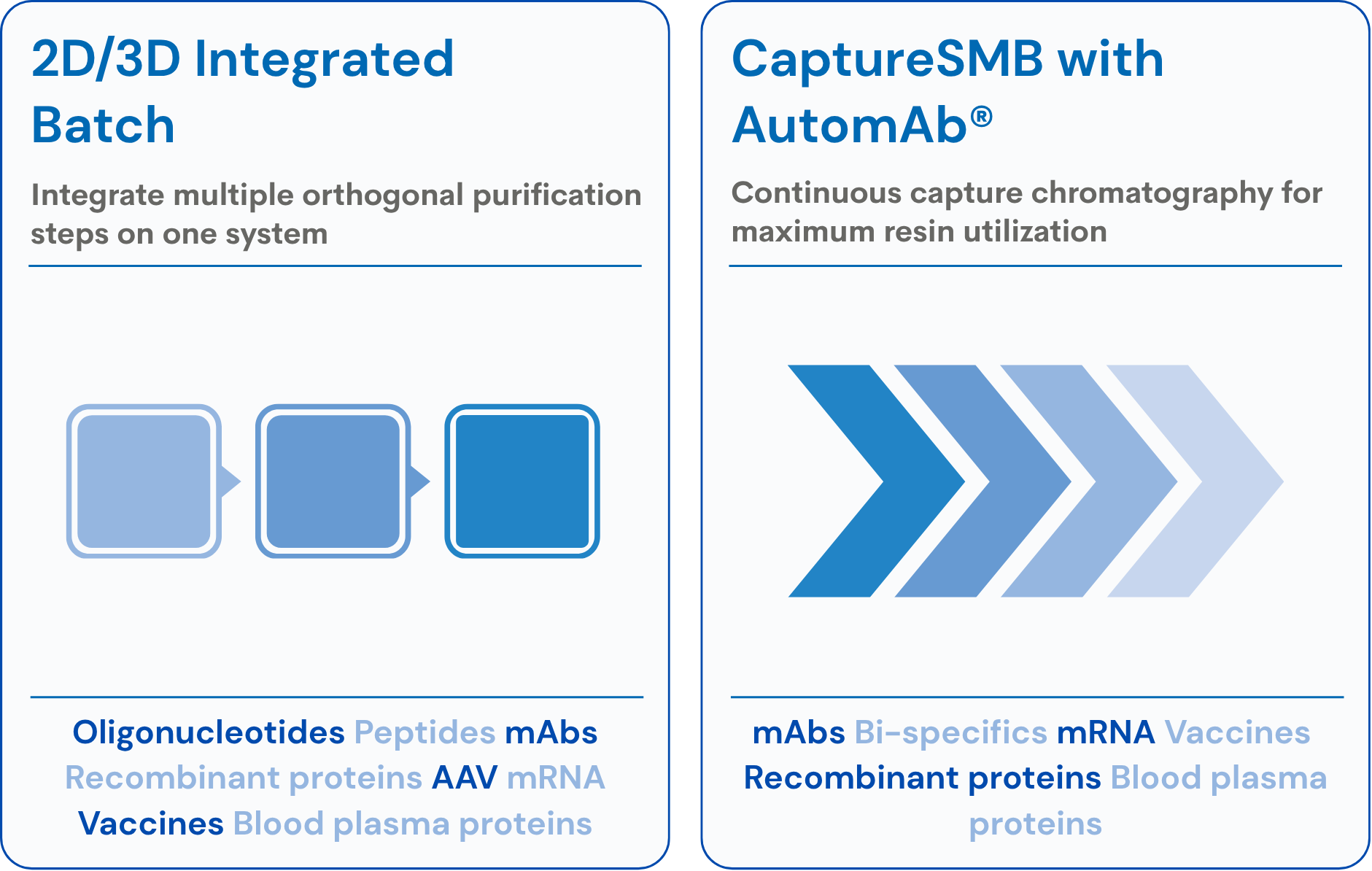
One System, Multiple Processes
Batch AND continuous capture on a single sanitary skid:
| Mode | Technology |
|---|---|
| Batch | Single-column isocratic and gradient |
| 2D Integrated Batch | Two-step purification with in-line dilution (BID) |
| Continuous Capture | CaptureSMB® with AutomAb® |
- Switch seamlessly between batch and CaptureSMB® modes
- Buffer in-line dilution (BID) option for on-board pH adjustment
- Eliminates duplicate equipment — saves space and capital
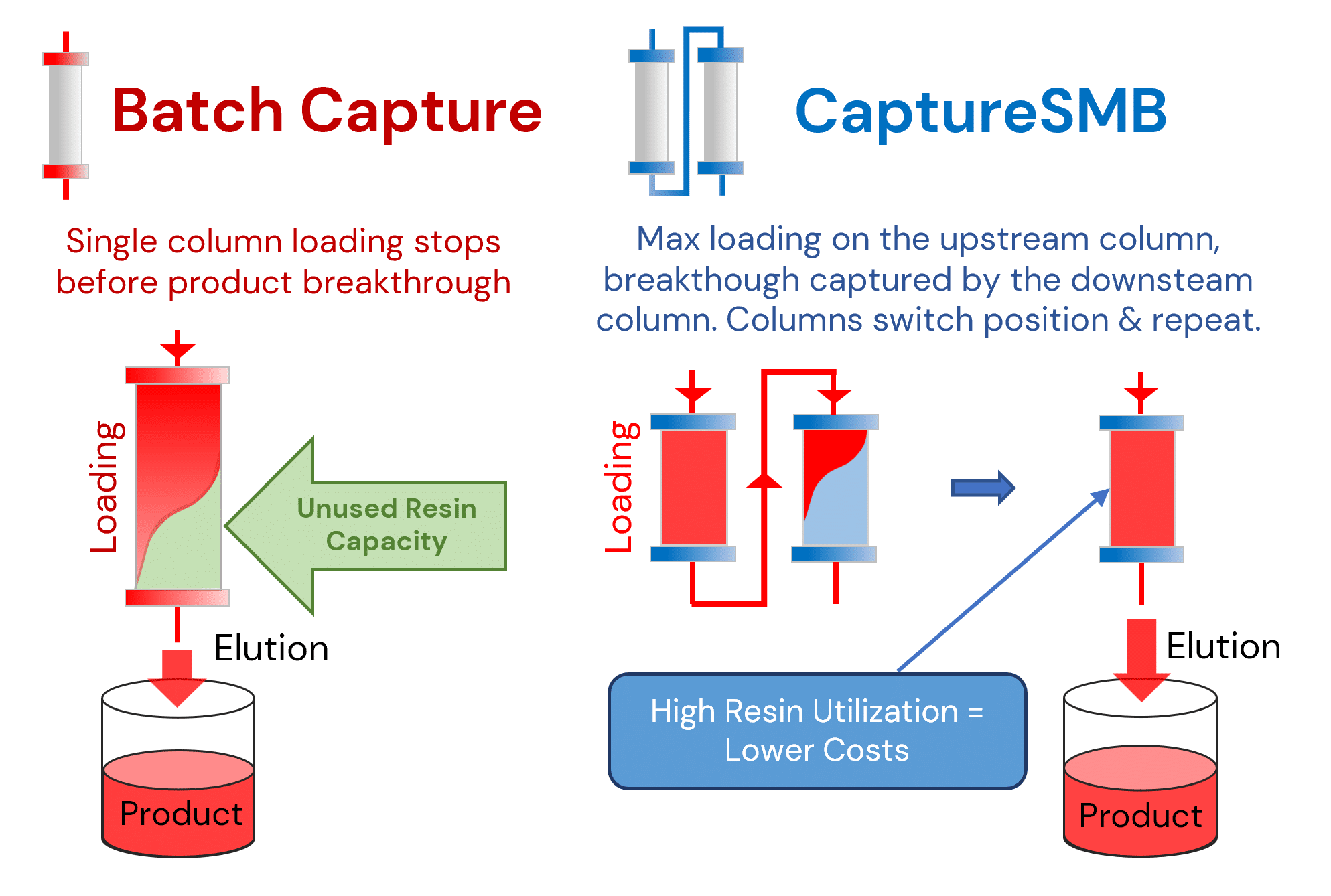
Patented CaptureSMB Technology
Twin-column continuous capture maximizing resin binding capacity:
| Metric | Value |
|---|---|
| Speed | Up to 3× faster than batch |
| Resin & buffer | Up to 50% reduction |
| Productivity | 2–4× vs. single-column batch |
- Two columns alternate loading and elution — continuous output
- Columns in series so the second captures breakthrough from the first
- Near-complete resin utilization eliminates re-chromatography
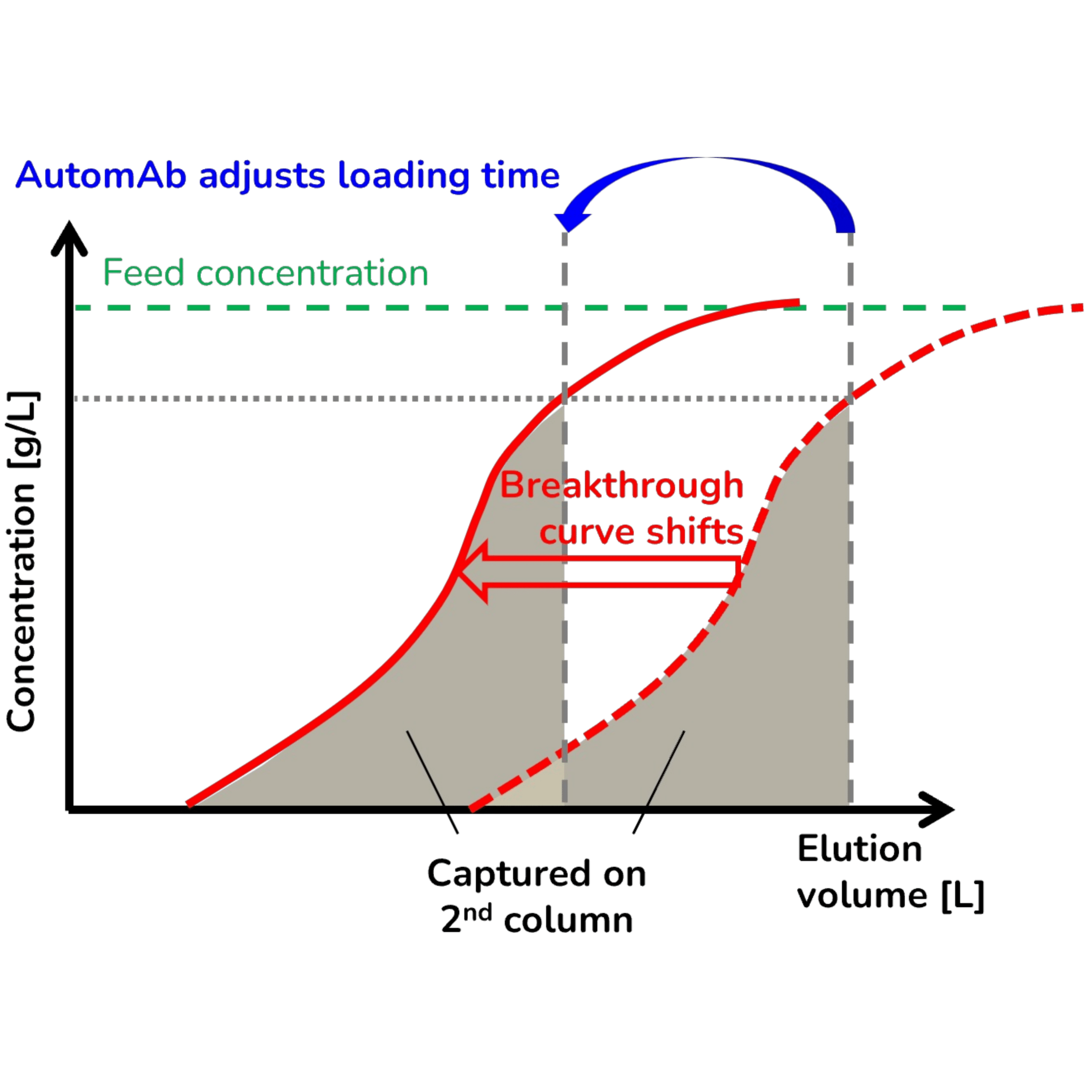
AutomAb® - Dynamic Process Control
AutomAb® monitors UV breakthrough in real time and adjusts column switching automatically:
- Detects product breakthrough and triggers switching at the optimal point
- Compensates for feed variability, column aging, and resin degradation
- Delivers consistent capture efficiency without operator intervention
- Enables robust, continuous 24/7 manufacturing
- Steady state reached after only one cycle with <1% variability
Comprehensive Application Range
Production-scale capture for biopharmaceuticals:
| Application | CaptureSMB® | Integrated Batch |
|---|---|---|
| mAbs & Antibody Variants | ✓ | ✓ |
| Viral Vectors (AAV) | ✓ | ✓ |
| Antibody-drug Conjugates | ✓ | ✓ |
| Recombinant Proteins | ✓ | ✓ |
- All processes run on the same sanitary hardware
- AutomAb® dynamic control across continuous operations
Scalable from Lab to Production
Part of the Contichrom® platform with established transfer protocols:
| System | Flow Rate | Column ID |
|---|---|---|
| CUBE 30/100 | 0.1–100 mL/min | 0.5–5 cm |
| TWIN LPLC 300 | 0.03 – 3.33 L/min | 10–20 cm |
| TWIN LPLC 500 | 0.08 – 8.33 L/min | 20–45 cm |
| TWIN LPLC 1000 | 0.15 – 18.9 L/min | 30–60 cm |
| TWIN LPLC 2000 | 0.5 – 36 L/min | 60–100 cm |
- Scale-up from CUBE at over 100× verified and published
- Same CaptureSMB® and AutomAb® control at every scale
Sanitary Design for Bioprocessing
Purpose-built for biopharmaceutical manufacturing environments:
- Fully sanitary flow path with drainable design and CIP compatibility
- ASME-BPE compliant stainless steel welds on all product contact surfaces
- SS 316L or Hastelloy flow path materials
- SF5 spec product contact tubing (≤ 20 Ra EP)
- Designed for NaOH 1M CIP and compatible with single-use aseptic interfaces
- Maximum operating pressure of 7.5 bar — optimal for affinity and LP resins
Designed for GMP Compliance
Built to meet stringent biopharmaceutical manufacturing requirements:
- Developed under the GAMP 5 framework
- 21 CFR Part 11 compliant software with audit trail
- Comprehensive Turnover Package (TOP) with ASME-BPE compliant welds
- USP Class VI materials and FDA-approved seals
- UL-approved panels, CE marking including PED compliance
TOP includes: MTRs, pressure test reports, passivation reports, weld documentation, as-built drawings.
Production-Grade Hardware
A complete skid-mounted system with mobile frame:
- 2× Pumps (base) — LEWA ecodos triplex with rupture detection
- 3rd Pump (BID option) — Buffer in-line dilution
- Dual UV Detectors — 4-Channel Adjustable wavelength (200-600 nm), 0.5 mm path length
- 2× Coriolis flow meters + 8× pressure transmitters
- pH, conductivity & temperature — Post-column standard
- Air sensors — Pre-pump and pre-column
- Pneumatic valves — Sanitary design, fast-acting at low pressure
- 24″ HMI — Industrial touchscreen with FactoryTalk View SE
CIP-Ready Design
The Contichrom® TWIN LPLC – Capture is designed for cleaning-in-place:
- Dedicated CIP inlet and outlet manifolds integrated into the valve network
- Fully drainable flow path design minimizes carryover between campaigns
- Compatible with NaOH 1M CIP protocols
- Optional single-use aseptic interface for minimally classified suites
- Reduces turnaround time and supports multi-product facility operation
How it works

Sanitary Hardware for GMP Biopharmaceutical Capture Chromatography
The Contichrom TWIN LPLC – Capture is a complete sanitary skid-mounted unit for GMP manufacturing:
- Mobile frame with levelers and non-marking caster wheels
- 316L stainless steel frame with mechanically polished welds
- Sanitary flow path designed for drainability and CIP
- Two column positions for CaptureSMB and single-column batch
- Operating temperature: 4–25 °C ambient, 4–40 °C process
LEWA Ecodos Triplex Pumps
Industry-leading LEWA ecodos triplex pumps for precision and reliability:
| Pump | Role |
|---|---|
| Pump A | Feed delivery / Load buffer |
| Pump B | Elution / Wash / Equilibration |
| Pump C (optional) | Buffer in-line dilution (BID) |
- Triplex configuration for nearly pulse-free flow
- Sandwich diaphragm with rupture detection alarm
- Automatic stroke adjustment for optimal performance
- PTFE diaphragms — USP Class VI certified, FDA approved
- Base system includes 2 pumps; 3rd pump available with BID option
Pneumatic Valve Network
Sanitary pneumatic valves — fast-acting at low pressure:
- Inlet Selection Valves — Multiple ports per pump for automated buffer/feed switching
- Column Switching Valves — For CaptureSMB interconnected and parallel configurations
- Fraction Outlet Valves — Product and waste routing with drain connection
- CIP Manifolds — Dedicated inlet and outlet for cleaning-in-place
- Materials: 316L SS body, PTFE/EPDM diaphragm, SF5 spec product contact (≤ 20 Ra EP)
- Sanitary design
Dual Wavelength UV Detectors
Two post-column UV sensors for real-time monitoring and AutomAb® control:
| Parameter | Specification |
|---|---|
| Type | 4-channel, multi-wavelength DAD, 200–600 nm |
| Path length | 0.5 mm path (exchangeable for 0.1, 0.3, or 1 mm) |
- Provides the UV breakthrough signals that enables AutomAb® dynamic process control
- Critical for consistent CaptureSMB® capture efficiency
In-Line Process Sensors
Comprehensive real-time monitoring:
| Sensor | Quantity | Specification |
|---|---|---|
| Coriolis Flow Meters | 2 | Flow monitoring and control |
| Pressure Transmitters | 8 | 0–13.8 bar, ±1% FS |
| pH | Post-column (x2) | 0–13 pH, ±0.15 unit |
| Conductivity | Post-column (x2) | 0–200 mS/cm, ±1% of measured value |
| Temperature | Per column | 0–100 °C, ±0.25 °C at 25 °C |
| Air Sensors | Pre-pump + pre-column | Ultrasonic detection |
Column Management
Flexible column configurations for production scale:
- 2 column positions for CaptureSMB® twin-column operation
- Column ID range: 10–100 cm (depending on model)
- Low-pressure operation Max 7.5 bar — optimized for affinity resins
- Sanitary tri-clamp connections for column attachment
- Single-column batch mode available
Human-Machine Interface (HMI)
System control via industrial PC running FactoryTalk View SE:
| Component | Specification |
|---|---|
| Touchscreen | 24″ industrial HMI, panel-mounted |
| Industrial PC | Preloaded with control software |
| OS | Windows 11 Professional |
- Real-time process visualization, recipe management, batch reporting
GMP-Grade Flow Path
Every product-contact surface selected for biopharmaceutical compatibility:
| Component | Materials |
|---|---|
| Product Contact Tubing | 316L SS or Hastelloy, ≤ 20 Ra EP (SF5 spec) |
| Non-Product Contact Tubing | 316L SS, ≤ 20 Ra MP (SF1 spec) |
| Process Valves | 316L SS body, PTFE/EPDM diaphragm |
| Pump Diaphragms | PTFE (USP Class VI, FDA approved) |
- ASME-BPE compliant welds, drainable design, CIP-compatible
- Surface roughness < 0.8 µm Ra on wetted metallic parts
PLC-Based Control System
Industrial automation platform:
| Component | Specification |
|---|---|
| PLC Platform | Rockwell CompactLogix |
| HMI Runtime | FactoryTalk View SE |
| Process Control | CaptureSMB with AutomAb® |
| Database | Microsoft SQL Server |
| Historian | YMC Server Historian |
| Reports | YMC Server Reports (auto-generated) |
| Remote Access | HMS Ewon module (client-enabled) |
Optional Hardware
| Option | Specification |
|---|---|
| Buffer In-line Dilution (BID) | Pump C + inlet valves + static mixer + sensors |
| Additional Inlet Valves | Up to 6 per pump |
| Additional Outlet Valves | Up to 5 fraction + 1 waste |
| Filter Housings | Single or dual with valve manifolds |
| Bubble Traps | 2 with valve manifold and vent valves |
| CIP Manifolds | Aseptic single-use bag connection |
| Software Customizations | Handshake with upstream/downstream equipment, additional control loops, external valve/equipment control |
Process Capabilities

Continuous Capture for Biopharmaceutical Production
The Contichrom TWIN LPLC – Capture is optimized for twin-column continuous capture:
- CaptureSMB® with AutomAb® — Continuous capture
- 2D Integrated Batch — Two-step purification with in-line dilution (BID option)
- Batch — Single-column isocratic and gradient operation
- All accessible from a single sanitary GMP-ready platform

CaptureSMB® with AutomAb® — Continuous Capture
Twin-column continuous capture maximizing resin binding capacity:
- Two identical columns alternate between loading and elution
- Columns in series — second column captures breakthrough from the first
- AutomAb® detects UV breakthrough and triggers column switching
- Near-complete utilization of resin binding capacity
- Continuous feed processing eliminates batch size limitations
- Steady state after one cycle with <1% variability
Target: Affinity capture steps (Protein A, mixed-mode) Molecules: mAbs, antibody variants, viral vectors (AAV), ADCs, recombinant proteins
CaptureSMB® — Performance Gains
| Metric | Result |
|---|---|
| Processing speed | Up to 3× faster than batch |
| Protein A resin consumption | Up to 50% reduction |
| Buffer consumption | Up to 50% reduction |
| Productivity | 2–4× higher vs. single-column batch |
| Intermediate storage | Reduced — continuous output eliminates hold steps |
| Process control | AutomAb® enables continuous 24/7 operation |
| Scale-up variability | <1% — steady state after one cycle |
One column loads while the other elutes — continuous output, maximum resin utilization, consistent quality.
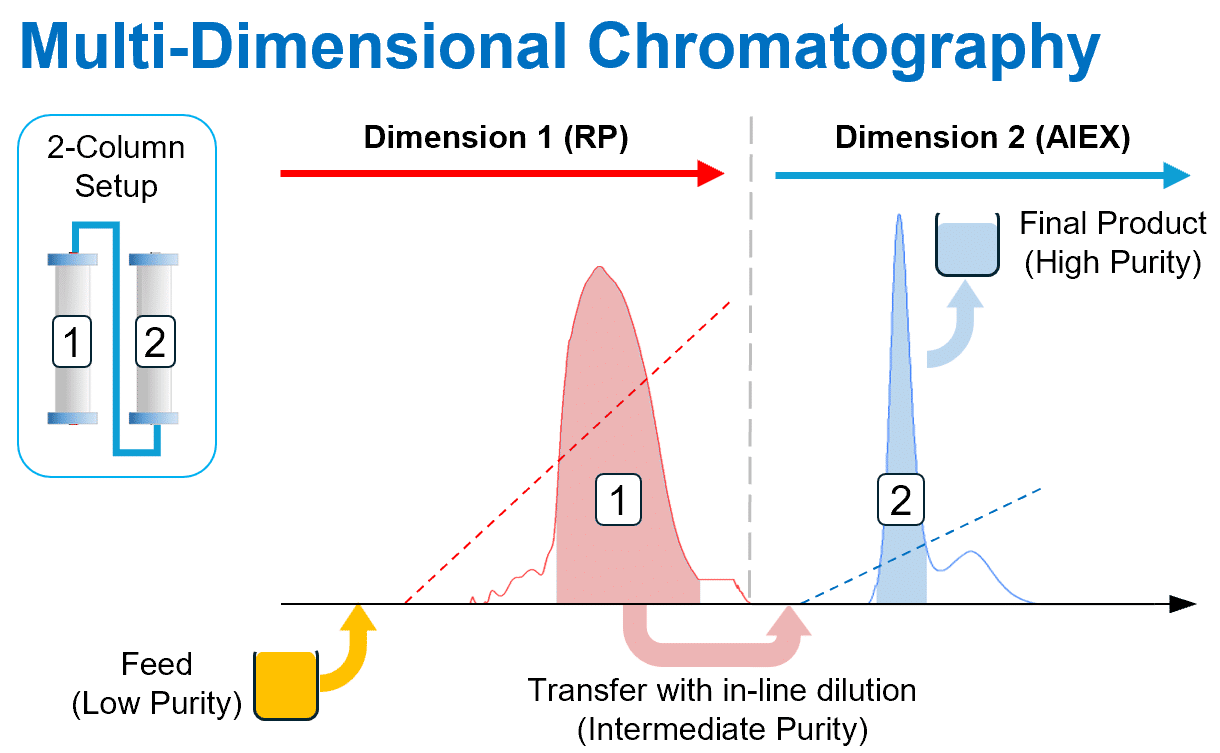
Integrated Batch — Two-Step Purification
Couple two chromatography steps without intermediate handling:
- Parallel or in-series column configurations
- Two chromatography modes run consecutively (e.g., IEX then HIC)
- On-board buffer in-line dilution (BID option) for pH adjustment
- Eliminates intermediate hold tanks and manual handling
- Reduces process time, QC burden, and facility footprint
Molecules: mAbs, antibody variants, viral vectors (AAV), ADCs, recombinant proteins
Batch Chromatography
Single-column batch purification:
| Mode | Applications |
|---|---|
| Isocratic | Affinity capture, SEC, desalting |
| Gradient | IEX, HIC, mixed-mode polishing |
- Linear gradient from 5% to 95% in batch mode
- Multiple fraction collection outlets
- Full UV/flow/pressure/pH/conductivity monitoring
Buffer In-line Dilution (BID)
On-board dilution of buffer concentrates and CIP solutions:
- Third pump (Pump C) delivers dilution buffer at controlled ratio
- Static mixer ensures homogeneous blending
- Enables pH adjustment between capture and polish steps
- Supports sequential processing for pool-less multi-step chromatography
Requires: BID option (Pump C, inlet valves, static mixer, sensors)
Regulatory Compliance & Documentation
Certifications
- UL-approved electrical panels
- CE marking available including PED compliance
- Developed under GAMP 5 framework
- Complies with FDA 21 CFR Part 11 regulations
Standards & Directives
- ASME-BPE for all stainless steel product contact welds
- FDA-approved USP Class VI materials and seals
- UL-approved electrical panels
- CE marking with PED compliance
Materials & Documentation
- All wetted materials selected for biopharmaceutical compatibility and chemical resistance
- Full traceability with MTRs and certificates of compliance
- Product contact surface roughness: < 0.8 µm Ra (SF5 spec: ≤ 20 Ra EP)
- Turnover Package (TOP) with comprehensive QA/QC documentation
TOP includes: MTRs, certificates of compliance, pressure test reports, passivation reports, weld documentation (logs, inspection reports, procedures, coupon logs, isometrics), slope verification, operation and maintenance manuals, as-built drawings (PDF and native CAD/Solidworks).
Specifications
System Models
| Parameter | TWIN 300 | TWIN 500 | TWIN 1000 | TWIN 2000 |
|---|---|---|---|---|
| Flow Rate | 0.03 – 3.33 L/min | 0.08 – 8.33 L/min | 0.15 – 18.9 L/min | 0.5 – 36 L/min |
| Column ID | 10–20 cm | 20–45 cm | 30–60 cm | 60–100 cm |
| Max Operating Pressure | 7.5 bar | 7.5 bar | 7.5 bar | 7.5 bar |
| Best For | Pilot / clinical | Mid-scale GMP | Large-scale GMP | Maximum capacity |
General Specifications
| Specification | Value |
|---|---|
| Max Operating Pressure | 7.5 bar (0.75 MPa / 109 psi) |
| Operating Temperature | 4–25 °C ambient; 4–40 °C process |
| Design | Sanitary, CIP-compatible (NaOH 1M) |
| Wetted Materials | SS 316L or Hastelloy, PTFE, EPDM (USP Class VI) |
| Surface Finish | < 0.8 µm Ra (product contact), SF5 spec: ≤ 20 Ra EP |
| Control System | Rockwell CompactLogix PLC |
| Processes | Batch, Integrated Batch, CaptureSMB® |
Physical Specifications
| Model | Length | Width | Height | Approx. Weight |
|---|---|---|---|---|
| TWIN 300 | 218 cm | 127 cm | 203 cm | 2494 kg |
| TWIN 500 | 299 cm | 165 cm | 198 cm | 3055 kg |
| TWIN 1000 (Pump) | 360 cm | 140 cm | 230 cm | 4455 kg |
| TWIN 1000 (Process) | 140 cm | 90 cm | 200 cm | 1364 kg |
| TWIN 2000 (Pump) | 381 cm | 152 cm | 230 cm | 4763 kg |
| TWIN 2000 (Process) | 147 cm | 101 cm | 200 cm | 1543 kg |
Pumps (LEWA ecodos)
| Configuration | 2× triplex diaphragm pumps (3× with BID option) |
| Diaphragm | Sandwich PTFE with rupture detection |
| Materials | USP Class VI certified, FDA approved |
| Features | Automatic stroke adjustment, nearly pulse-free flow |
Detectors & Sensors
| UV Detection | 4-channel, multi-wavelength DAD, 200–600 nm, 0.5 mm path (exchangeable for 0.1, 0.3, or 1 mm) |
| Flow Meters (×2) | Coriolis |
| Pressure (×8) | Transmitters throughout flow path, 0–13.8 bar, ±1% FS |
| pH | On waste and collection outlets, 0–13, ±0.15 unit |
| Conductivity | On waste and collection outlets, 0–200 mS/cm, ±1% of measured value |
| Temperature | Post-column, 0–100 °C, ±0.25 °C at 25 °C |
| Air Sensors | Pre-pump and pre-column (ultrasonic) |
Valves & Flow Path
| Valve Actuation | Pneumatic diaphragm valves |
| Valve Materials | 316L SS body, PTFE/EPDM diaphragm |
| Product Contact | SF5 spec: ≤ 20 Ra EP |
| Non-Product Contact | SF1 spec: ≤ 20 Ra MP |
| Column Connections | Sanitary tri-clamp |
| CIP | Dedicated inlet and outlet manifolds |
Software & Automation
| PLC Platform | Rockwell CompactLogix |
| HMI Runtime | FactoryTalk View SE |
| Process Control | CaptureSMB® with AutomAb® |
| Database | Microsoft SQL Server |
| Historian | YMC Server Historian |
| Reports | YMC Server Reports (auto-generated) |
| Remote Access | HMS Ewon module (client-enabled) |
| Data Integrity | 21 CFR Part 11, audit trail, electronic signatures |
User Interface
| HMI | 24″ industrial touchscreen, panel-mounted |
| Industrial PC | Preloaded with control software |
| OS | Windows 11 Professional |
Optional Modules
| Option | Specification |
|---|---|
| Buffer In-line Dilution (BID) | Pump C + inlet valves + static mixer + sensors |
| Additional Inlet Valves | Up to 6 per pump |
| Additional Outlet Valves | Up to 5 fraction + 1 waste |
| Filter Housings | Single or dual with valve manifolds |
| Bubble Traps | 2 with valve manifold and vent valves |
| CIP Manifolds | Aseptic single-use bag connection |
| Software Customizations | Handshake with upstream/downstream equipment, additional control loops, external valve/equipment control |
Technologies

Single-column purification with full method flexibility — gradient, isocratic, step elution. The foundation for process development and the starting point for continuous workflows.
Single-column purification with full method flexibility — gradient, isocratic, step elution. The foundation for process development and the starting point for continuous workflows.

Automate sequential purification across multiple columns and chemistries in a single run. ChromIQ® handles fraction transfer, timing, and method switching.
Automate sequential purification across multiple columns and chemistries in a single run. ChromIQ® handles fraction transfer, timing, and method switching.

Twin-column continuous loading maximizes resin utilization and throughput. AutomAb® dynamically controls breakthrough — adapting in real time to feed variability.
Twin-column continuous loading maximizes resin utilization and throughput. AutomAb® dynamically controls breakthrough — adapting in real time to feed variability.

Real-time UV-based control that adapts your continuous process to feed and column variability — eliminating the need for fixed, conservative operating parameters.
Real-time UV-based control that adapts your continuous process to feed and column variability — eliminating the need for fixed, conservative operating parameters.
Applications

CaptureSMB® with AutomAb® for mAbs & antibody variants: maximize productivity and resin utilization, while slashing buffer consumption.
CaptureSMB® with AutomAb® for mAbs & antibody variants: maximize productivity and resin utilization, while slashing buffer consumption.
From capture through polishing, Contichrom® integrates multiple chromatography steps into automated 2D/3D workflows — reducing hands-on time and intermediate handling.
From capture through polishing, Contichrom® integrates multiple chromatography steps into automated 2D/3D workflows — reducing hands-on time and intermediate handling.

MCSGP with AutoPeak® is ideally suited for DAR control, achieving precise drug-antibody ratios with higher yield than batch chromatography.
MCSGP with AutoPeak® is ideally suited for DAR control, achieving precise drug-antibody ratios with higher yield than batch chromatography.
Resources
- All Media Types
- Peer-Reviewed Publications
Continued insights into virus clearance validation across continuous capture chromatography, Biotechnol. Bioeng.
Additional viral validation studies for CaptureSMB validate a surrogate batch model that accurately predicts virus clearance in continuous capture operations.
Model-based process development of continuous chromatography for antibody capture: A case study with twin-column system, J. Chromatogr. A
Model-based approaches for twin-column continuous capture process design establish systematic methodology for CaptureSMB implementation.
Virus Clearance Validation across Continuous Capture Chromatography. Biotechnol. Bioeng.
Viral validation studies conducted under cGLP guidelines for CaptureSMB demonstrate comparable virus clearance to batch processing and establish regulatory-compliant validation approaches.
Service & Support

See Contichrom® in action. On-site, at our facility, or remote — get a first-hand look at what our chromatography systems and software can do.
See Contichrom® in action. On-site, at our facility, or remote — get a first-hand look at what our chromatography systems and software can do.

Hands-on operator training, advanced application workshops, and expert consulting — customised to your team's experience and your specific Contichrom® applications.
Hands-on operator training, advanced application workshops, and expert consulting — customised to your team's experience and your specific Contichrom® applications.

Calibrated mechanistic models built from your batch data — simulate and optimize batch, MCSGP, or CaptureSMB® processes computationally, before running a single experiment.
Calibrated mechanistic models built from your batch data — simulate and optimize batch, MCSGP, or CaptureSMB® processes computationally, before running a single experiment.

Try a Contichrom® system in your own lab before committing to a purchase. Full technical support and training included — evaluate the technology on your terms.
Try a Contichrom® system in your own lab before committing to a purchase. Full technical support and training included — evaluate the technology on your terms.

Quantified performance comparison. Run your molecule on our Contichrom® systems and benchmark CaptureSMB® or MCSGP against your existing batch process.
Quantified performance comparison. Run your molecule on our Contichrom® systems and benchmark CaptureSMB® or MCSGP against your existing batch process.

Preventive maintenance subscriptions and on-site repair services to keep your Contichrom® running at peak performance. Remote technical support available worldwide.
Preventive maintenance subscriptions and on-site repair services to keep your Contichrom® running at peak performance. Remote technical support available worldwide.
FAQ
A complete Contichrom® TWIN LPLC – Capture system includes:
- Complete sanitary skid assembly with mobile frame (levelers and casters)
- 2× LEWA ecodos triplex pumps with sandwich diaphragms and rupture detection
- Full inlet and outlet pneumatic valve network (sanitary design)
- Dual wavelength UV detectors (280 nm, 303 nm)
- Post-column pH and conductivity sensors
- 2× Coriolis flow meters + 8× pressure transmitters
- Pre-pump and pre-column air sensors
- CIP inlet and outlet manifolds
- 24″ HMI with industrial PC and control software preloaded
- Rockwell CompactLogix PLC control system
- Comprehensive TOP documentation package
- Factory Acceptance Testing (FAT) — up to 5 days
- One week on-site startup support and training
Customer provides: Columns, filter elements, chromatography resin, external piping, utilities connections.
| Feature | TWIN HPLC | TWIN LPLC Capture | TWIN LPLC Polishing |
|---|---|---|---|
| Design | Industrial (non-sanitary) | Sanitary | Sanitary |
| Pressure | 80 bar | 7.5 bar (20 bar opt.) | 7.5 bar (20 bar opt.) |
| Core Process | MCSGP (polishing) | CaptureSMB® (capture) | MCSGP (polishing) |
| HP Valves | YMC fast-acting | Standard pneumatic | Standard pneumatic |
| Pumps | 4× LEWA ecoflow | 2× (3×) LEWA ecodos | 4× LEWA ecodos |
| Molecules | Synthetic peptides, oligos | Biologics (mAbs, AAV, ADCs) | Biologics (mAbs, AAV, ADCs) |
The TWIN LPLC – Capture is the right choice for affinity capture of biopharmaceuticals where sanitary design and CIP capability are required.
| Model | Column ID | Flow Rate | Best For |
|---|---|---|---|
| TWIN 300 | 10–20 cm | 0.03 – 3.33 L/min | Pilot production, clinical manufacturing |
| TWIN 500 | 20–45 cm | 0.08 – 8.33 L/min | Mid-scale commercial production |
| TWIN 1000 | 30–60 cm | 0.15 – 18.9 L/min | Large-scale commercial production |
| TWIN 2000 | 60–100 cm | 0.5 – 36 L/min | Maximum capacity production |
Contact YMC to discuss your throughput requirements and optimal system selection.
- Floor space: Dimensions vary by model (contact YMC for GA drawings)
- Power: Model-dependent (contact YMC for specifications)
- Utilities: Process air, water, compressed air, drain, network connection
- Ambient temperature: 4–25 °C
- Process temperature: 4–40 °C
Note: System must be uncrated, leveled, and connected to utilities before YMC on-site work begins.
- Methods developed on Contichrom CUBE transfer directly to the TWIN LPLC
- Same CaptureSMB® principles and AutomAb® control at both scales
- Scale-up at over 100× verified and published
- Steady state after one cycle with <1% variability
- YMC ChromaCon provides process transfer support
- FAT — Up to 5 days at YMC facility
- On-site startup — One week including commissioning and system walkdown
- Training — 1 full day or 2 half-days covering operation and maintenance
- Remote support — HMS Ewon remote access for troubleshooting and training
- Warranty: 12 months from installation or 18 months from shipment (whichever first)
- Global service: YMC operations worldwide, LEWA support in 80 countries
- Remote access: Built-in capability for automation troubleshooting
- Components: Non-proprietary with global distribution for local acquisition
- Maintenance: No special tools required for standard maintenance
Yes — customization options include:
- Buffer in-line dilution (BID) with additional pump
- Additional inlet valves (up to 6 on Pump A, B, or C)
- Additional outlet/fraction valves (up to 5 fraction + 1 waste)
- Pre-column analytics (pH and conductivity)
- Single or dual filter housings
- Bubble traps
- CIP manifolds for aseptic single-use bag connection
Contact YMC ChromaCon for custom configuration pricing.
Overview

GMP-Ready Continuous Capture for Biopharmaceutical Manufacturing
The Contichrom® TWIN LPLC – Capture is a sanitary chromatography system for continuous capture of biologics from pilot to large-scale GMP manufacturing.
- Twin-column continuous capture (CaptureSMB® with AutomAb®)
- Up to 3× faster processing, 50% less resin and buffer vs. batch
- Sanitary design with CIP capability for biopharmaceutical production
- SS 316L or Hastelloy flow paths, 7.5 bar maximum operating pressure
- 21 CFR Part 11 compliant software with full audit trail

One System, Multiple Processes
Batch AND continuous capture on a single sanitary skid:
| Mode | Technology |
|---|---|
| Batch | Single-column isocratic and gradient |
| 2D Integrated Batch | Two-step purification with in-line dilution (BID) |
| Continuous Capture | CaptureSMB® with AutomAb® |
- Switch seamlessly between batch and CaptureSMB® modes
- Buffer in-line dilution (BID) option for on-board pH adjustment
- Eliminates duplicate equipment — saves space and capital

Patented CaptureSMB Technology
Twin-column continuous capture maximizing resin binding capacity:
| Metric | Value |
|---|---|
| Speed | Up to 3× faster than batch |
| Resin & buffer | Up to 50% reduction |
| Productivity | 2–4× vs. single-column batch |
- Two columns alternate loading and elution — continuous output
- Columns in series so the second captures breakthrough from the first
- Near-complete resin utilization eliminates re-chromatography

AutomAb® - Dynamic Process Control
AutomAb® monitors UV breakthrough in real time and adjusts column switching automatically:
- Detects product breakthrough and triggers switching at the optimal point
- Compensates for feed variability, column aging, and resin degradation
- Delivers consistent capture efficiency without operator intervention
- Enables robust, continuous 24/7 manufacturing
- Steady state reached after only one cycle with <1% variability
Comprehensive Application Range
Production-scale capture for biopharmaceuticals:
| Application | CaptureSMB® | Integrated Batch |
|---|---|---|
| mAbs & Antibody Variants | ✓ | ✓ |
| Viral Vectors (AAV) | ✓ | ✓ |
| Antibody-drug Conjugates | ✓ | ✓ |
| Recombinant Proteins | ✓ | ✓ |
- All processes run on the same sanitary hardware
- AutomAb® dynamic control across continuous operations
Scalable from Lab to Production
Part of the Contichrom® platform with established transfer protocols:
| System | Flow Rate | Column ID |
|---|---|---|
| CUBE 30/100 | 0.1–100 mL/min | 0.5–5 cm |
| TWIN LPLC 300 | 0.03 – 3.33 L/min | 10–20 cm |
| TWIN LPLC 500 | 0.08 – 8.33 L/min | 20–45 cm |
| TWIN LPLC 1000 | 0.15 – 18.9 L/min | 30–60 cm |
| TWIN LPLC 2000 | 0.5 – 36 L/min | 60–100 cm |
- Scale-up from CUBE at over 100× verified and published
- Same CaptureSMB® and AutomAb® control at every scale
Sanitary Design for Bioprocessing
Purpose-built for biopharmaceutical manufacturing environments:
- Fully sanitary flow path with drainable design and CIP compatibility
- ASME-BPE compliant stainless steel welds on all product contact surfaces
- SS 316L or Hastelloy flow path materials
- SF5 spec product contact tubing (≤ 20 Ra EP)
- Designed for NaOH 1M CIP and compatible with single-use aseptic interfaces
- Maximum operating pressure of 7.5 bar — optimal for affinity and LP resins
Designed for GMP Compliance
Built to meet stringent biopharmaceutical manufacturing requirements:
- Developed under the GAMP 5 framework
- 21 CFR Part 11 compliant software with audit trail
- Comprehensive Turnover Package (TOP) with ASME-BPE compliant welds
- USP Class VI materials and FDA-approved seals
- UL-approved panels, CE marking including PED compliance
TOP includes: MTRs, pressure test reports, passivation reports, weld documentation, as-built drawings.
Production-Grade Hardware
A complete skid-mounted system with mobile frame:
- 2× Pumps (base) — LEWA ecodos triplex with rupture detection
- 3rd Pump (BID option) — Buffer in-line dilution
- Dual UV Detectors — 4-Channel Adjustable wavelength (200-600 nm), 0.5 mm path length
- 2× Coriolis flow meters + 8× pressure transmitters
- pH, conductivity & temperature — Post-column standard
- Air sensors — Pre-pump and pre-column
- Pneumatic valves — Sanitary design, fast-acting at low pressure
- 24″ HMI — Industrial touchscreen with FactoryTalk View SE
CIP-Ready Design
The Contichrom® TWIN LPLC – Capture is designed for cleaning-in-place:
- Dedicated CIP inlet and outlet manifolds integrated into the valve network
- Fully drainable flow path design minimizes carryover between campaigns
- Compatible with NaOH 1M CIP protocols
- Optional single-use aseptic interface for minimally classified suites
- Reduces turnaround time and supports multi-product facility operation
How it works

Sanitary Hardware for GMP Biopharmaceutical Capture Chromatography
The Contichrom TWIN LPLC – Capture is a complete sanitary skid-mounted unit for GMP manufacturing:
- Mobile frame with levelers and non-marking caster wheels
- 316L stainless steel frame with mechanically polished welds
- Sanitary flow path designed for drainability and CIP
- Two column positions for CaptureSMB and single-column batch
- Operating temperature: 4–25 °C ambient, 4–40 °C process
LEWA Ecodos Triplex Pumps
Industry-leading LEWA ecodos triplex pumps for precision and reliability:
| Pump | Role |
|---|---|
| Pump A | Feed delivery / Load buffer |
| Pump B | Elution / Wash / Equilibration |
| Pump C (optional) | Buffer in-line dilution (BID) |
- Triplex configuration for nearly pulse-free flow
- Sandwich diaphragm with rupture detection alarm
- Automatic stroke adjustment for optimal performance
- PTFE diaphragms — USP Class VI certified, FDA approved
- Base system includes 2 pumps; 3rd pump available with BID option
Pneumatic Valve Network
Sanitary pneumatic valves — fast-acting at low pressure:
- Inlet Selection Valves — Multiple ports per pump for automated buffer/feed switching
- Column Switching Valves — For CaptureSMB interconnected and parallel configurations
- Fraction Outlet Valves — Product and waste routing with drain connection
- CIP Manifolds — Dedicated inlet and outlet for cleaning-in-place
- Materials: 316L SS body, PTFE/EPDM diaphragm, SF5 spec product contact (≤ 20 Ra EP)
- Sanitary design
Dual Wavelength UV Detectors
Two post-column UV sensors for real-time monitoring and AutomAb® control:
| Parameter | Specification |
|---|---|
| Type | 4-channel, multi-wavelength DAD, 200–600 nm |
| Path length | 0.5 mm path (exchangeable for 0.1, 0.3, or 1 mm) |
- Provides the UV breakthrough signals that enables AutomAb® dynamic process control
- Critical for consistent CaptureSMB® capture efficiency
In-Line Process Sensors
Comprehensive real-time monitoring:
| Sensor | Quantity | Specification |
|---|---|---|
| Coriolis Flow Meters | 2 | Flow monitoring and control |
| Pressure Transmitters | 8 | 0–13.8 bar, ±1% FS |
| pH | Post-column (x2) | 0–13 pH, ±0.15 unit |
| Conductivity | Post-column (x2) | 0–200 mS/cm, ±1% of measured value |
| Temperature | Per column | 0–100 °C, ±0.25 °C at 25 °C |
| Air Sensors | Pre-pump + pre-column | Ultrasonic detection |
Column Management
Flexible column configurations for production scale:
- 2 column positions for CaptureSMB® twin-column operation
- Column ID range: 10–100 cm (depending on model)
- Low-pressure operation Max 7.5 bar — optimized for affinity resins
- Sanitary tri-clamp connections for column attachment
- Single-column batch mode available
Human-Machine Interface (HMI)
System control via industrial PC running FactoryTalk View SE:
| Component | Specification |
|---|---|
| Touchscreen | 24″ industrial HMI, panel-mounted |
| Industrial PC | Preloaded with control software |
| OS | Windows 11 Professional |
- Real-time process visualization, recipe management, batch reporting
GMP-Grade Flow Path
Every product-contact surface selected for biopharmaceutical compatibility:
| Component | Materials |
|---|---|
| Product Contact Tubing | 316L SS or Hastelloy, ≤ 20 Ra EP (SF5 spec) |
| Non-Product Contact Tubing | 316L SS, ≤ 20 Ra MP (SF1 spec) |
| Process Valves | 316L SS body, PTFE/EPDM diaphragm |
| Pump Diaphragms | PTFE (USP Class VI, FDA approved) |
- ASME-BPE compliant welds, drainable design, CIP-compatible
- Surface roughness < 0.8 µm Ra on wetted metallic parts
PLC-Based Control System
Industrial automation platform:
| Component | Specification |
|---|---|
| PLC Platform | Rockwell CompactLogix |
| HMI Runtime | FactoryTalk View SE |
| Process Control | CaptureSMB with AutomAb® |
| Database | Microsoft SQL Server |
| Historian | YMC Server Historian |
| Reports | YMC Server Reports (auto-generated) |
| Remote Access | HMS Ewon module (client-enabled) |
Optional Hardware
| Option | Specification |
|---|---|
| Buffer In-line Dilution (BID) | Pump C + inlet valves + static mixer + sensors |
| Additional Inlet Valves | Up to 6 per pump |
| Additional Outlet Valves | Up to 5 fraction + 1 waste |
| Filter Housings | Single or dual with valve manifolds |
| Bubble Traps | 2 with valve manifold and vent valves |
| CIP Manifolds | Aseptic single-use bag connection |
| Software Customizations | Handshake with upstream/downstream equipment, additional control loops, external valve/equipment control |
Process Capabilities

Continuous Capture for Biopharmaceutical Production
The Contichrom TWIN LPLC – Capture is optimized for twin-column continuous capture:
- CaptureSMB® with AutomAb® — Continuous capture
- 2D Integrated Batch — Two-step purification with in-line dilution (BID option)
- Batch — Single-column isocratic and gradient operation
- All accessible from a single sanitary GMP-ready platform

CaptureSMB® with AutomAb® — Continuous Capture
Twin-column continuous capture maximizing resin binding capacity:
- Two identical columns alternate between loading and elution
- Columns in series — second column captures breakthrough from the first
- AutomAb® detects UV breakthrough and triggers column switching
- Near-complete utilization of resin binding capacity
- Continuous feed processing eliminates batch size limitations
- Steady state after one cycle with <1% variability
Target: Affinity capture steps (Protein A, mixed-mode) Molecules: mAbs, antibody variants, viral vectors (AAV), ADCs, recombinant proteins
CaptureSMB® — Performance Gains
| Metric | Result |
|---|---|
| Processing speed | Up to 3× faster than batch |
| Protein A resin consumption | Up to 50% reduction |
| Buffer consumption | Up to 50% reduction |
| Productivity | 2–4× higher vs. single-column batch |
| Intermediate storage | Reduced — continuous output eliminates hold steps |
| Process control | AutomAb® enables continuous 24/7 operation |
| Scale-up variability | <1% — steady state after one cycle |
One column loads while the other elutes — continuous output, maximum resin utilization, consistent quality.

Integrated Batch — Two-Step Purification
Couple two chromatography steps without intermediate handling:
- Parallel or in-series column configurations
- Two chromatography modes run consecutively (e.g., IEX then HIC)
- On-board buffer in-line dilution (BID option) for pH adjustment
- Eliminates intermediate hold tanks and manual handling
- Reduces process time, QC burden, and facility footprint
Molecules: mAbs, antibody variants, viral vectors (AAV), ADCs, recombinant proteins
Batch Chromatography
Single-column batch purification:
| Mode | Applications |
|---|---|
| Isocratic | Affinity capture, SEC, desalting |
| Gradient | IEX, HIC, mixed-mode polishing |
- Linear gradient from 5% to 95% in batch mode
- Multiple fraction collection outlets
- Full UV/flow/pressure/pH/conductivity monitoring
Buffer In-line Dilution (BID)
On-board dilution of buffer concentrates and CIP solutions:
- Third pump (Pump C) delivers dilution buffer at controlled ratio
- Static mixer ensures homogeneous blending
- Enables pH adjustment between capture and polish steps
- Supports sequential processing for pool-less multi-step chromatography
Requires: BID option (Pump C, inlet valves, static mixer, sensors)
Regulatory Compliance & Documentation
Certifications
- UL-approved electrical panels
- CE marking available including PED compliance
- Developed under GAMP 5 framework
- Complies with FDA 21 CFR Part 11 regulations
Standards & Directives
- ASME-BPE for all stainless steel product contact welds
- FDA-approved USP Class VI materials and seals
- UL-approved electrical panels
- CE marking with PED compliance
Materials & Documentation
- All wetted materials selected for biopharmaceutical compatibility and chemical resistance
- Full traceability with MTRs and certificates of compliance
- Product contact surface roughness: < 0.8 µm Ra (SF5 spec: ≤ 20 Ra EP)
- Turnover Package (TOP) with comprehensive QA/QC documentation
TOP includes: MTRs, certificates of compliance, pressure test reports, passivation reports, weld documentation (logs, inspection reports, procedures, coupon logs, isometrics), slope verification, operation and maintenance manuals, as-built drawings (PDF and native CAD/Solidworks).
Specifications
System Models
| Parameter | TWIN 300 | TWIN 500 | TWIN 1000 | TWIN 2000 |
|---|---|---|---|---|
| Flow Rate | 0.03 – 3.33 L/min | 0.08 – 8.33 L/min | 0.15 – 18.9 L/min | 0.5 – 36 L/min |
| Column ID | 10–20 cm | 20–45 cm | 30–60 cm | 60–100 cm |
| Max Operating Pressure | 7.5 bar | 7.5 bar | 7.5 bar | 7.5 bar |
| Best For | Pilot / clinical | Mid-scale GMP | Large-scale GMP | Maximum capacity |
General Specifications
| Specification | Value |
|---|---|
| Max Operating Pressure | 7.5 bar (0.75 MPa / 109 psi) |
| Operating Temperature | 4–25 °C ambient; 4–40 °C process |
| Design | Sanitary, CIP-compatible (NaOH 1M) |
| Wetted Materials | SS 316L or Hastelloy, PTFE, EPDM (USP Class VI) |
| Surface Finish | < 0.8 µm Ra (product contact), SF5 spec: ≤ 20 Ra EP |
| Control System | Rockwell CompactLogix PLC |
| Processes | Batch, Integrated Batch, CaptureSMB® |
Physical Specifications
| Model | Length | Width | Height | Approx. Weight |
|---|---|---|---|---|
| TWIN 300 | 218 cm | 127 cm | 203 cm | 2494 kg |
| TWIN 500 | 299 cm | 165 cm | 198 cm | 3055 kg |
| TWIN 1000 (Pump) | 360 cm | 140 cm | 230 cm | 4455 kg |
| TWIN 1000 (Process) | 140 cm | 90 cm | 200 cm | 1364 kg |
| TWIN 2000 (Pump) | 381 cm | 152 cm | 230 cm | 4763 kg |
| TWIN 2000 (Process) | 147 cm | 101 cm | 200 cm | 1543 kg |
Pumps (LEWA ecodos)
| Configuration | 2× triplex diaphragm pumps (3× with BID option) |
| Diaphragm | Sandwich PTFE with rupture detection |
| Materials | USP Class VI certified, FDA approved |
| Features | Automatic stroke adjustment, nearly pulse-free flow |
Detectors & Sensors
| UV Detection | 4-channel, multi-wavelength DAD, 200–600 nm, 0.5 mm path (exchangeable for 0.1, 0.3, or 1 mm) |
| Flow Meters (×2) | Coriolis |
| Pressure (×8) | Transmitters throughout flow path, 0–13.8 bar, ±1% FS |
| pH | On waste and collection outlets, 0–13, ±0.15 unit |
| Conductivity | On waste and collection outlets, 0–200 mS/cm, ±1% of measured value |
| Temperature | Post-column, 0–100 °C, ±0.25 °C at 25 °C |
| Air Sensors | Pre-pump and pre-column (ultrasonic) |
Valves & Flow Path
| Valve Actuation | Pneumatic diaphragm valves |
| Valve Materials | 316L SS body, PTFE/EPDM diaphragm |
| Product Contact | SF5 spec: ≤ 20 Ra EP |
| Non-Product Contact | SF1 spec: ≤ 20 Ra MP |
| Column Connections | Sanitary tri-clamp |
| CIP | Dedicated inlet and outlet manifolds |
Software & Automation
| PLC Platform | Rockwell CompactLogix |
| HMI Runtime | FactoryTalk View SE |
| Process Control | CaptureSMB® with AutomAb® |
| Database | Microsoft SQL Server |
| Historian | YMC Server Historian |
| Reports | YMC Server Reports (auto-generated) |
| Remote Access | HMS Ewon module (client-enabled) |
| Data Integrity | 21 CFR Part 11, audit trail, electronic signatures |
User Interface
| HMI | 24″ industrial touchscreen, panel-mounted |
| Industrial PC | Preloaded with control software |
| OS | Windows 11 Professional |
Optional Modules
| Option | Specification |
|---|---|
| Buffer In-line Dilution (BID) | Pump C + inlet valves + static mixer + sensors |
| Additional Inlet Valves | Up to 6 per pump |
| Additional Outlet Valves | Up to 5 fraction + 1 waste |
| Filter Housings | Single or dual with valve manifolds |
| Bubble Traps | 2 with valve manifold and vent valves |
| CIP Manifolds | Aseptic single-use bag connection |
| Software Customizations | Handshake with upstream/downstream equipment, additional control loops, external valve/equipment control |
Technologies

Single-column purification with full method flexibility — gradient, isocratic, step elution. The foundation for process development and the starting point for continuous workflows.
Single-column purification with full method flexibility — gradient, isocratic, step elution. The foundation for process development and the starting point for continuous workflows.

Automate sequential purification across multiple columns and chemistries in a single run. ChromIQ® handles fraction transfer, timing, and method switching.
Automate sequential purification across multiple columns and chemistries in a single run. ChromIQ® handles fraction transfer, timing, and method switching.

Twin-column continuous loading maximizes resin utilization and throughput. AutomAb® dynamically controls breakthrough — adapting in real time to feed variability.
Twin-column continuous loading maximizes resin utilization and throughput. AutomAb® dynamically controls breakthrough — adapting in real time to feed variability.

Real-time UV-based control that adapts your continuous process to feed and column variability — eliminating the need for fixed, conservative operating parameters.
Real-time UV-based control that adapts your continuous process to feed and column variability — eliminating the need for fixed, conservative operating parameters.
Applications

CaptureSMB® with AutomAb® for mAbs & antibody variants: maximize productivity and resin utilization, while slashing buffer consumption.
CaptureSMB® with AutomAb® for mAbs & antibody variants: maximize productivity and resin utilization, while slashing buffer consumption.

From capture through polishing, Contichrom® integrates multiple chromatography steps into automated 2D/3D workflows — reducing hands-on time and intermediate handling.
From capture through polishing, Contichrom® integrates multiple chromatography steps into automated 2D/3D workflows — reducing hands-on time and intermediate handling.

MCSGP with AutoPeak® is ideally suited for DAR control, achieving precise drug-antibody ratios with higher yield than batch chromatography.
MCSGP with AutoPeak® is ideally suited for DAR control, achieving precise drug-antibody ratios with higher yield than batch chromatography.
Resources
- All Media Types
- Peer-Reviewed Publications
Continued insights into virus clearance validation across continuous capture chromatography, Biotechnol. Bioeng.
Additional viral validation studies for CaptureSMB validate a surrogate batch model that accurately predicts virus clearance in continuous capture operations.
Model-based process development of continuous chromatography for antibody capture: A case study with twin-column system, J. Chromatogr. A
Model-based approaches for twin-column continuous capture process design establish systematic methodology for CaptureSMB implementation.
Virus Clearance Validation across Continuous Capture Chromatography. Biotechnol. Bioeng.
Viral validation studies conducted under cGLP guidelines for CaptureSMB demonstrate comparable virus clearance to batch processing and establish regulatory-compliant validation approaches.
Service & Support

See Contichrom® in action. On-site, at our facility, or remote — get a first-hand look at what our chromatography systems and software can do.
See Contichrom® in action. On-site, at our facility, or remote — get a first-hand look at what our chromatography systems and software can do.

Hands-on operator training, advanced application workshops, and expert consulting — customised to your team's experience and your specific Contichrom® applications.
Hands-on operator training, advanced application workshops, and expert consulting — customised to your team's experience and your specific Contichrom® applications.

Calibrated mechanistic models built from your batch data — simulate and optimize batch, MCSGP, or CaptureSMB® processes computationally, before running a single experiment.
Calibrated mechanistic models built from your batch data — simulate and optimize batch, MCSGP, or CaptureSMB® processes computationally, before running a single experiment.

Try a Contichrom® system in your own lab before committing to a purchase. Full technical support and training included — evaluate the technology on your terms.
Try a Contichrom® system in your own lab before committing to a purchase. Full technical support and training included — evaluate the technology on your terms.

Quantified performance comparison. Run your molecule on our Contichrom® systems and benchmark CaptureSMB® or MCSGP against your existing batch process.
Quantified performance comparison. Run your molecule on our Contichrom® systems and benchmark CaptureSMB® or MCSGP against your existing batch process.

Preventive maintenance subscriptions and on-site repair services to keep your Contichrom® running at peak performance. Remote technical support available worldwide.
Preventive maintenance subscriptions and on-site repair services to keep your Contichrom® running at peak performance. Remote technical support available worldwide.
FAQ
A complete Contichrom® TWIN LPLC – Capture system includes:
- Complete sanitary skid assembly with mobile frame (levelers and casters)
- 2× LEWA ecodos triplex pumps with sandwich diaphragms and rupture detection
- Full inlet and outlet pneumatic valve network (sanitary design)
- Dual wavelength UV detectors (280 nm, 303 nm)
- Post-column pH and conductivity sensors
- 2× Coriolis flow meters + 8× pressure transmitters
- Pre-pump and pre-column air sensors
- CIP inlet and outlet manifolds
- 24″ HMI with industrial PC and control software preloaded
- Rockwell CompactLogix PLC control system
- Comprehensive TOP documentation package
- Factory Acceptance Testing (FAT) — up to 5 days
- One week on-site startup support and training
Customer provides: Columns, filter elements, chromatography resin, external piping, utilities connections.
| Feature | TWIN HPLC | TWIN LPLC Capture | TWIN LPLC Polishing |
|---|---|---|---|
| Design | Industrial (non-sanitary) | Sanitary | Sanitary |
| Pressure | 80 bar | 7.5 bar (20 bar opt.) | 7.5 bar (20 bar opt.) |
| Core Process | MCSGP (polishing) | CaptureSMB® (capture) | MCSGP (polishing) |
| HP Valves | YMC fast-acting | Standard pneumatic | Standard pneumatic |
| Pumps | 4× LEWA ecoflow | 2× (3×) LEWA ecodos | 4× LEWA ecodos |
| Molecules | Synthetic peptides, oligos | Biologics (mAbs, AAV, ADCs) | Biologics (mAbs, AAV, ADCs) |
The TWIN LPLC – Capture is the right choice for affinity capture of biopharmaceuticals where sanitary design and CIP capability are required.
| Model | Column ID | Flow Rate | Best For |
|---|---|---|---|
| TWIN 300 | 10–20 cm | 0.03 – 3.33 L/min | Pilot production, clinical manufacturing |
| TWIN 500 | 20–45 cm | 0.08 – 8.33 L/min | Mid-scale commercial production |
| TWIN 1000 | 30–60 cm | 0.15 – 18.9 L/min | Large-scale commercial production |
| TWIN 2000 | 60–100 cm | 0.5 – 36 L/min | Maximum capacity production |
Contact YMC to discuss your throughput requirements and optimal system selection.
- Floor space: Dimensions vary by model (contact YMC for GA drawings)
- Power: Model-dependent (contact YMC for specifications)
- Utilities: Process air, water, compressed air, drain, network connection
- Ambient temperature: 4–25 °C
- Process temperature: 4–40 °C
Note: System must be uncrated, leveled, and connected to utilities before YMC on-site work begins.
- Methods developed on Contichrom CUBE transfer directly to the TWIN LPLC
- Same CaptureSMB® principles and AutomAb® control at both scales
- Scale-up at over 100× verified and published
- Steady state after one cycle with <1% variability
- YMC ChromaCon provides process transfer support
- FAT — Up to 5 days at YMC facility
- On-site startup — One week including commissioning and system walkdown
- Training — 1 full day or 2 half-days covering operation and maintenance
- Remote support — HMS Ewon remote access for troubleshooting and training
- Warranty: 12 months from installation or 18 months from shipment (whichever first)
- Global service: YMC operations worldwide, LEWA support in 80 countries
- Remote access: Built-in capability for automation troubleshooting
- Components: Non-proprietary with global distribution for local acquisition
- Maintenance: No special tools required for standard maintenance
Yes — customization options include:
- Buffer in-line dilution (BID) with additional pump
- Additional inlet valves (up to 6 on Pump A, B, or C)
- Additional outlet/fraction valves (up to 5 fraction + 1 waste)
- Pre-column analytics (pH and conductivity)
- Single or dual filter housings
- Bubble traps
- CIP manifolds for aseptic single-use bag connection
Contact YMC ChromaCon for custom configuration pricing.
Overview

GMP-Ready Continuous Capture for Biopharmaceutical Manufacturing
The Contichrom® TWIN LPLC – Capture is a sanitary chromatography system for continuous capture of biologics from pilot to large-scale GMP manufacturing.
- Twin-column continuous capture (CaptureSMB® with AutomAb®)
- Up to 3× faster processing, 50% less resin and buffer vs. batch
- Sanitary design with CIP capability for biopharmaceutical production
- SS 316L or Hastelloy flow paths, 7.5 bar maximum operating pressure
- 21 CFR Part 11 compliant software with full audit trail

One System, Multiple Processes
Batch AND continuous capture on a single sanitary skid:
| Mode | Technology |
|---|---|
| Batch | Single-column isocratic and gradient |
| 2D Integrated Batch | Two-step purification with in-line dilution (BID) |
| Continuous Capture | CaptureSMB® with AutomAb® |
- Switch seamlessly between batch and CaptureSMB® modes
- Buffer in-line dilution (BID) option for on-board pH adjustment
- Eliminates duplicate equipment — saves space and capital

Patented CaptureSMB Technology
Twin-column continuous capture maximizing resin binding capacity:
| Metric | Value |
|---|---|
| Speed | Up to 3× faster than batch |
| Resin & buffer | Up to 50% reduction |
| Productivity | 2–4× vs. single-column batch |
- Two columns alternate loading and elution — continuous output
- Columns in series so the second captures breakthrough from the first
- Near-complete resin utilization eliminates re-chromatography

AutomAb® - Dynamic Process Control
AutomAb® monitors UV breakthrough in real time and adjusts column switching automatically:
- Detects product breakthrough and triggers switching at the optimal point
- Compensates for feed variability, column aging, and resin degradation
- Delivers consistent capture efficiency without operator intervention
- Enables robust, continuous 24/7 manufacturing
- Steady state reached after only one cycle with <1% variability
Comprehensive Application Range
Production-scale capture for biopharmaceuticals:
| Application | CaptureSMB® | Integrated Batch |
|---|---|---|
| mAbs & Antibody Variants | ✓ | ✓ |
| Viral Vectors (AAV) | ✓ | ✓ |
| Antibody-drug Conjugates | ✓ | ✓ |
| Recombinant Proteins | ✓ | ✓ |
- All processes run on the same sanitary hardware
- AutomAb® dynamic control across continuous operations
Scalable from Lab to Production
Part of the Contichrom® platform with established transfer protocols:
| System | Flow Rate | Column ID |
|---|---|---|
| CUBE 30/100 | 0.1–100 mL/min | 0.5–5 cm |
| TWIN LPLC 300 | 0.03 – 3.33 L/min | 10–20 cm |
| TWIN LPLC 500 | 0.08 – 8.33 L/min | 20–45 cm |
| TWIN LPLC 1000 | 0.15 – 18.9 L/min | 30–60 cm |
| TWIN LPLC 2000 | 0.5 – 36 L/min | 60–100 cm |
- Scale-up from CUBE at over 100× verified and published
- Same CaptureSMB® and AutomAb® control at every scale
Sanitary Design for Bioprocessing
Purpose-built for biopharmaceutical manufacturing environments:
- Fully sanitary flow path with drainable design and CIP compatibility
- ASME-BPE compliant stainless steel welds on all product contact surfaces
- SS 316L or Hastelloy flow path materials
- SF5 spec product contact tubing (≤ 20 Ra EP)
- Designed for NaOH 1M CIP and compatible with single-use aseptic interfaces
- Maximum operating pressure of 7.5 bar — optimal for affinity and LP resins
Designed for GMP Compliance
Built to meet stringent biopharmaceutical manufacturing requirements:
- Developed under the GAMP 5 framework
- 21 CFR Part 11 compliant software with audit trail
- Comprehensive Turnover Package (TOP) with ASME-BPE compliant welds
- USP Class VI materials and FDA-approved seals
- UL-approved panels, CE marking including PED compliance
TOP includes: MTRs, pressure test reports, passivation reports, weld documentation, as-built drawings.
Production-Grade Hardware
A complete skid-mounted system with mobile frame:
- 2× Pumps (base) — LEWA ecodos triplex with rupture detection
- 3rd Pump (BID option) — Buffer in-line dilution
- Dual UV Detectors — 4-Channel Adjustable wavelength (200-600 nm), 0.5 mm path length
- 2× Coriolis flow meters + 8× pressure transmitters
- pH, conductivity & temperature — Post-column standard
- Air sensors — Pre-pump and pre-column
- Pneumatic valves — Sanitary design, fast-acting at low pressure
- 24″ HMI — Industrial touchscreen with FactoryTalk View SE
CIP-Ready Design
The Contichrom® TWIN LPLC – Capture is designed for cleaning-in-place:
- Dedicated CIP inlet and outlet manifolds integrated into the valve network
- Fully drainable flow path design minimizes carryover between campaigns
- Compatible with NaOH 1M CIP protocols
- Optional single-use aseptic interface for minimally classified suites
- Reduces turnaround time and supports multi-product facility operation
How it works

Sanitary Hardware for GMP Biopharmaceutical Capture Chromatography
The Contichrom TWIN LPLC – Capture is a complete sanitary skid-mounted unit for GMP manufacturing:
- Mobile frame with levelers and non-marking caster wheels
- 316L stainless steel frame with mechanically polished welds
- Sanitary flow path designed for drainability and CIP
- Two column positions for CaptureSMB and single-column batch
- Operating temperature: 4–25 °C ambient, 4–40 °C process
LEWA Ecodos Triplex Pumps
Industry-leading LEWA ecodos triplex pumps for precision and reliability:
| Pump | Role |
|---|---|
| Pump A | Feed delivery / Load buffer |
| Pump B | Elution / Wash / Equilibration |
| Pump C (optional) | Buffer in-line dilution (BID) |
- Triplex configuration for nearly pulse-free flow
- Sandwich diaphragm with rupture detection alarm
- Automatic stroke adjustment for optimal performance
- PTFE diaphragms — USP Class VI certified, FDA approved
- Base system includes 2 pumps; 3rd pump available with BID option
Pneumatic Valve Network
Sanitary pneumatic valves — fast-acting at low pressure:
- Inlet Selection Valves — Multiple ports per pump for automated buffer/feed switching
- Column Switching Valves — For CaptureSMB interconnected and parallel configurations
- Fraction Outlet Valves — Product and waste routing with drain connection
- CIP Manifolds — Dedicated inlet and outlet for cleaning-in-place
- Materials: 316L SS body, PTFE/EPDM diaphragm, SF5 spec product contact (≤ 20 Ra EP)
- Sanitary design
Dual Wavelength UV Detectors
Two post-column UV sensors for real-time monitoring and AutomAb® control:
| Parameter | Specification |
|---|---|
| Type | 4-channel, multi-wavelength DAD, 200–600 nm |
| Path length | 0.5 mm path (exchangeable for 0.1, 0.3, or 1 mm) |
- Provides the UV breakthrough signals that enables AutomAb® dynamic process control
- Critical for consistent CaptureSMB® capture efficiency
In-Line Process Sensors
Comprehensive real-time monitoring:
| Sensor | Quantity | Specification |
|---|---|---|
| Coriolis Flow Meters | 2 | Flow monitoring and control |
| Pressure Transmitters | 8 | 0–13.8 bar, ±1% FS |
| pH | Post-column (x2) | 0–13 pH, ±0.15 unit |
| Conductivity | Post-column (x2) | 0–200 mS/cm, ±1% of measured value |
| Temperature | Per column | 0–100 °C, ±0.25 °C at 25 °C |
| Air Sensors | Pre-pump + pre-column | Ultrasonic detection |
Column Management
Flexible column configurations for production scale:
- 2 column positions for CaptureSMB® twin-column operation
- Column ID range: 10–100 cm (depending on model)
- Low-pressure operation Max 7.5 bar — optimized for affinity resins
- Sanitary tri-clamp connections for column attachment
- Single-column batch mode available
Human-Machine Interface (HMI)
System control via industrial PC running FactoryTalk View SE:
| Component | Specification |
|---|---|
| Touchscreen | 24″ industrial HMI, panel-mounted |
| Industrial PC | Preloaded with control software |
| OS | Windows 11 Professional |
- Real-time process visualization, recipe management, batch reporting
GMP-Grade Flow Path
Every product-contact surface selected for biopharmaceutical compatibility:
| Component | Materials |
|---|---|
| Product Contact Tubing | 316L SS or Hastelloy, ≤ 20 Ra EP (SF5 spec) |
| Non-Product Contact Tubing | 316L SS, ≤ 20 Ra MP (SF1 spec) |
| Process Valves | 316L SS body, PTFE/EPDM diaphragm |
| Pump Diaphragms | PTFE (USP Class VI, FDA approved) |
- ASME-BPE compliant welds, drainable design, CIP-compatible
- Surface roughness < 0.8 µm Ra on wetted metallic parts
PLC-Based Control System
Industrial automation platform:
| Component | Specification |
|---|---|
| PLC Platform | Rockwell CompactLogix |
| HMI Runtime | FactoryTalk View SE |
| Process Control | CaptureSMB with AutomAb® |
| Database | Microsoft SQL Server |
| Historian | YMC Server Historian |
| Reports | YMC Server Reports (auto-generated) |
| Remote Access | HMS Ewon module (client-enabled) |
Optional Hardware
| Option | Specification |
|---|---|
| Buffer In-line Dilution (BID) | Pump C + inlet valves + static mixer + sensors |
| Additional Inlet Valves | Up to 6 per pump |
| Additional Outlet Valves | Up to 5 fraction + 1 waste |
| Filter Housings | Single or dual with valve manifolds |
| Bubble Traps | 2 with valve manifold and vent valves |
| CIP Manifolds | Aseptic single-use bag connection |
| Software Customizations | Handshake with upstream/downstream equipment, additional control loops, external valve/equipment control |
Process Capabilities

Continuous Capture for Biopharmaceutical Production
The Contichrom TWIN LPLC – Capture is optimized for twin-column continuous capture:
- CaptureSMB® with AutomAb® — Continuous capture
- 2D Integrated Batch — Two-step purification with in-line dilution (BID option)
- Batch — Single-column isocratic and gradient operation
- All accessible from a single sanitary GMP-ready platform

CaptureSMB® with AutomAb® — Continuous Capture
Twin-column continuous capture maximizing resin binding capacity:
- Two identical columns alternate between loading and elution
- Columns in series — second column captures breakthrough from the first
- AutomAb® detects UV breakthrough and triggers column switching
- Near-complete utilization of resin binding capacity
- Continuous feed processing eliminates batch size limitations
- Steady state after one cycle with <1% variability
Target: Affinity capture steps (Protein A, mixed-mode) Molecules: mAbs, antibody variants, viral vectors (AAV), ADCs, recombinant proteins
CaptureSMB® — Performance Gains
| Metric | Result |
|---|---|
| Processing speed | Up to 3× faster than batch |
| Protein A resin consumption | Up to 50% reduction |
| Buffer consumption | Up to 50% reduction |
| Productivity | 2–4× higher vs. single-column batch |
| Intermediate storage | Reduced — continuous output eliminates hold steps |
| Process control | AutomAb® enables continuous 24/7 operation |
| Scale-up variability | <1% — steady state after one cycle |
One column loads while the other elutes — continuous output, maximum resin utilization, consistent quality.

Integrated Batch — Two-Step Purification
Couple two chromatography steps without intermediate handling:
- Parallel or in-series column configurations
- Two chromatography modes run consecutively (e.g., IEX then HIC)
- On-board buffer in-line dilution (BID option) for pH adjustment
- Eliminates intermediate hold tanks and manual handling
- Reduces process time, QC burden, and facility footprint
Molecules: mAbs, antibody variants, viral vectors (AAV), ADCs, recombinant proteins
Batch Chromatography
Single-column batch purification:
| Mode | Applications |
|---|---|
| Isocratic | Affinity capture, SEC, desalting |
| Gradient | IEX, HIC, mixed-mode polishing |
- Linear gradient from 5% to 95% in batch mode
- Multiple fraction collection outlets
- Full UV/flow/pressure/pH/conductivity monitoring
Buffer In-line Dilution (BID)
On-board dilution of buffer concentrates and CIP solutions:
- Third pump (Pump C) delivers dilution buffer at controlled ratio
- Static mixer ensures homogeneous blending
- Enables pH adjustment between capture and polish steps
- Supports sequential processing for pool-less multi-step chromatography
Requires: BID option (Pump C, inlet valves, static mixer, sensors)
Regulatory Compliance & Documentation
Certifications
- UL-approved electrical panels
- CE marking available including PED compliance
- Developed under GAMP 5 framework
- Complies with FDA 21 CFR Part 11 regulations
Standards & Directives
- ASME-BPE for all stainless steel product contact welds
- FDA-approved USP Class VI materials and seals
- UL-approved electrical panels
- CE marking with PED compliance
Materials & Documentation
- All wetted materials selected for biopharmaceutical compatibility and chemical resistance
- Full traceability with MTRs and certificates of compliance
- Product contact surface roughness: < 0.8 µm Ra (SF5 spec: ≤ 20 Ra EP)
- Turnover Package (TOP) with comprehensive QA/QC documentation
TOP includes: MTRs, certificates of compliance, pressure test reports, passivation reports, weld documentation (logs, inspection reports, procedures, coupon logs, isometrics), slope verification, operation and maintenance manuals, as-built drawings (PDF and native CAD/Solidworks).
Specifications
System Models
| Parameter | TWIN 300 | TWIN 500 | TWIN 1000 | TWIN 2000 |
|---|---|---|---|---|
| Flow Rate | 0.03 – 3.33 L/min | 0.08 – 8.33 L/min | 0.15 – 18.9 L/min | 0.5 – 36 L/min |
| Column ID | 10–20 cm | 20–45 cm | 30–60 cm | 60–100 cm |
| Max Operating Pressure | 7.5 bar | 7.5 bar | 7.5 bar | 7.5 bar |
| Best For | Pilot / clinical | Mid-scale GMP | Large-scale GMP | Maximum capacity |
General Specifications
| Specification | Value |
|---|---|
| Max Operating Pressure | 7.5 bar (0.75 MPa / 109 psi) |
| Operating Temperature | 4–25 °C ambient; 4–40 °C process |
| Design | Sanitary, CIP-compatible (NaOH 1M) |
| Wetted Materials | SS 316L or Hastelloy, PTFE, EPDM (USP Class VI) |
| Surface Finish | < 0.8 µm Ra (product contact), SF5 spec: ≤ 20 Ra EP |
| Control System | Rockwell CompactLogix PLC |
| Processes | Batch, Integrated Batch, CaptureSMB® |
Physical Specifications
| Model | Length | Width | Height | Approx. Weight |
|---|---|---|---|---|
| TWIN 300 | 218 cm | 127 cm | 203 cm | 2494 kg |
| TWIN 500 | 299 cm | 165 cm | 198 cm | 3055 kg |
| TWIN 1000 (Pump) | 360 cm | 140 cm | 230 cm | 4455 kg |
| TWIN 1000 (Process) | 140 cm | 90 cm | 200 cm | 1364 kg |
| TWIN 2000 (Pump) | 381 cm | 152 cm | 230 cm | 4763 kg |
| TWIN 2000 (Process) | 147 cm | 101 cm | 200 cm | 1543 kg |
Pumps (LEWA ecodos)
| Configuration | 2× triplex diaphragm pumps (3× with BID option) |
| Diaphragm | Sandwich PTFE with rupture detection |
| Materials | USP Class VI certified, FDA approved |
| Features | Automatic stroke adjustment, nearly pulse-free flow |
Detectors & Sensors
| UV Detection | 4-channel, multi-wavelength DAD, 200–600 nm, 0.5 mm path (exchangeable for 0.1, 0.3, or 1 mm) |
| Flow Meters (×2) | Coriolis |
| Pressure (×8) | Transmitters throughout flow path, 0–13.8 bar, ±1% FS |
| pH | On waste and collection outlets, 0–13, ±0.15 unit |
| Conductivity | On waste and collection outlets, 0–200 mS/cm, ±1% of measured value |
| Temperature | Post-column, 0–100 °C, ±0.25 °C at 25 °C |
| Air Sensors | Pre-pump and pre-column (ultrasonic) |
Valves & Flow Path
| Valve Actuation | Pneumatic diaphragm valves |
| Valve Materials | 316L SS body, PTFE/EPDM diaphragm |
| Product Contact | SF5 spec: ≤ 20 Ra EP |
| Non-Product Contact | SF1 spec: ≤ 20 Ra MP |
| Column Connections | Sanitary tri-clamp |
| CIP | Dedicated inlet and outlet manifolds |
Software & Automation
| PLC Platform | Rockwell CompactLogix |
| HMI Runtime | FactoryTalk View SE |
| Process Control | CaptureSMB® with AutomAb® |
| Database | Microsoft SQL Server |
| Historian | YMC Server Historian |
| Reports | YMC Server Reports (auto-generated) |
| Remote Access | HMS Ewon module (client-enabled) |
| Data Integrity | 21 CFR Part 11, audit trail, electronic signatures |
User Interface
| HMI | 24″ industrial touchscreen, panel-mounted |
| Industrial PC | Preloaded with control software |
| OS | Windows 11 Professional |
Optional Modules
| Option | Specification |
|---|---|
| Buffer In-line Dilution (BID) | Pump C + inlet valves + static mixer + sensors |
| Additional Inlet Valves | Up to 6 per pump |
| Additional Outlet Valves | Up to 5 fraction + 1 waste |
| Filter Housings | Single or dual with valve manifolds |
| Bubble Traps | 2 with valve manifold and vent valves |
| CIP Manifolds | Aseptic single-use bag connection |
| Software Customizations | Handshake with upstream/downstream equipment, additional control loops, external valve/equipment control |
Technologies

Single-column purification with full method flexibility — gradient, isocratic, step elution. The foundation for process development and the starting point for continuous workflows.
Single-column purification with full method flexibility — gradient, isocratic, step elution. The foundation for process development and the starting point for continuous workflows.

Automate sequential purification across multiple columns and chemistries in a single run. ChromIQ® handles fraction transfer, timing, and method switching.
Automate sequential purification across multiple columns and chemistries in a single run. ChromIQ® handles fraction transfer, timing, and method switching.

Twin-column continuous loading maximizes resin utilization and throughput. AutomAb® dynamically controls breakthrough — adapting in real time to feed variability.
Twin-column continuous loading maximizes resin utilization and throughput. AutomAb® dynamically controls breakthrough — adapting in real time to feed variability.

Real-time UV-based control that adapts your continuous process to feed and column variability — eliminating the need for fixed, conservative operating parameters.
Real-time UV-based control that adapts your continuous process to feed and column variability — eliminating the need for fixed, conservative operating parameters.
Applications

CaptureSMB® with AutomAb® for mAbs & antibody variants: maximize productivity and resin utilization, while slashing buffer consumption.
CaptureSMB® with AutomAb® for mAbs & antibody variants: maximize productivity and resin utilization, while slashing buffer consumption.

From capture through polishing, Contichrom® integrates multiple chromatography steps into automated 2D/3D workflows — reducing hands-on time and intermediate handling.
From capture through polishing, Contichrom® integrates multiple chromatography steps into automated 2D/3D workflows — reducing hands-on time and intermediate handling.

MCSGP with AutoPeak® is ideally suited for DAR control, achieving precise drug-antibody ratios with higher yield than batch chromatography.
MCSGP with AutoPeak® is ideally suited for DAR control, achieving precise drug-antibody ratios with higher yield than batch chromatography.
Resources
- All Media Types
- Peer-Reviewed Publications
Continued insights into virus clearance validation across continuous capture chromatography, Biotechnol. Bioeng.
Additional viral validation studies for CaptureSMB validate a surrogate batch model that accurately predicts virus clearance in continuous capture operations.
Model-based process development of continuous chromatography for antibody capture: A case study with twin-column system, J. Chromatogr. A
Model-based approaches for twin-column continuous capture process design establish systematic methodology for CaptureSMB implementation.
Virus Clearance Validation across Continuous Capture Chromatography. Biotechnol. Bioeng.
Viral validation studies conducted under cGLP guidelines for CaptureSMB demonstrate comparable virus clearance to batch processing and establish regulatory-compliant validation approaches.
Service & Support

See Contichrom® in action. On-site, at our facility, or remote — get a first-hand look at what our chromatography systems and software can do.
See Contichrom® in action. On-site, at our facility, or remote — get a first-hand look at what our chromatography systems and software can do.

Hands-on operator training, advanced application workshops, and expert consulting — customised to your team's experience and your specific Contichrom® applications.
Hands-on operator training, advanced application workshops, and expert consulting — customised to your team's experience and your specific Contichrom® applications.

Calibrated mechanistic models built from your batch data — simulate and optimize batch, MCSGP, or CaptureSMB® processes computationally, before running a single experiment.
Calibrated mechanistic models built from your batch data — simulate and optimize batch, MCSGP, or CaptureSMB® processes computationally, before running a single experiment.

Try a Contichrom® system in your own lab before committing to a purchase. Full technical support and training included — evaluate the technology on your terms.
Try a Contichrom® system in your own lab before committing to a purchase. Full technical support and training included — evaluate the technology on your terms.

Quantified performance comparison. Run your molecule on our Contichrom® systems and benchmark CaptureSMB® or MCSGP against your existing batch process.
Quantified performance comparison. Run your molecule on our Contichrom® systems and benchmark CaptureSMB® or MCSGP against your existing batch process.

Preventive maintenance subscriptions and on-site repair services to keep your Contichrom® running at peak performance. Remote technical support available worldwide.
Preventive maintenance subscriptions and on-site repair services to keep your Contichrom® running at peak performance. Remote technical support available worldwide.
FAQ
A complete Contichrom® TWIN LPLC – Capture system includes:
- Complete sanitary skid assembly with mobile frame (levelers and casters)
- 2× LEWA ecodos triplex pumps with sandwich diaphragms and rupture detection
- Full inlet and outlet pneumatic valve network (sanitary design)
- Dual wavelength UV detectors (280 nm, 303 nm)
- Post-column pH and conductivity sensors
- 2× Coriolis flow meters + 8× pressure transmitters
- Pre-pump and pre-column air sensors
- CIP inlet and outlet manifolds
- 24″ HMI with industrial PC and control software preloaded
- Rockwell CompactLogix PLC control system
- Comprehensive TOP documentation package
- Factory Acceptance Testing (FAT) — up to 5 days
- One week on-site startup support and training
Customer provides: Columns, filter elements, chromatography resin, external piping, utilities connections.
| Feature | TWIN HPLC | TWIN LPLC Capture | TWIN LPLC Polishing |
|---|---|---|---|
| Design | Industrial (non-sanitary) | Sanitary | Sanitary |
| Pressure | 80 bar | 7.5 bar (20 bar opt.) | 7.5 bar (20 bar opt.) |
| Core Process | MCSGP (polishing) | CaptureSMB® (capture) | MCSGP (polishing) |
| HP Valves | YMC fast-acting | Standard pneumatic | Standard pneumatic |
| Pumps | 4× LEWA ecoflow | 2× (3×) LEWA ecodos | 4× LEWA ecodos |
| Molecules | Synthetic peptides, oligos | Biologics (mAbs, AAV, ADCs) | Biologics (mAbs, AAV, ADCs) |
The TWIN LPLC – Capture is the right choice for affinity capture of biopharmaceuticals where sanitary design and CIP capability are required.
| Model | Column ID | Flow Rate | Best For |
|---|---|---|---|
| TWIN 300 | 10–20 cm | 0.03 – 3.33 L/min | Pilot production, clinical manufacturing |
| TWIN 500 | 20–45 cm | 0.08 – 8.33 L/min | Mid-scale commercial production |
| TWIN 1000 | 30–60 cm | 0.15 – 18.9 L/min | Large-scale commercial production |
| TWIN 2000 | 60–100 cm | 0.5 – 36 L/min | Maximum capacity production |
Contact YMC to discuss your throughput requirements and optimal system selection.
- Floor space: Dimensions vary by model (contact YMC for GA drawings)
- Power: Model-dependent (contact YMC for specifications)
- Utilities: Process air, water, compressed air, drain, network connection
- Ambient temperature: 4–25 °C
- Process temperature: 4–40 °C
Note: System must be uncrated, leveled, and connected to utilities before YMC on-site work begins.
- Methods developed on Contichrom CUBE transfer directly to the TWIN LPLC
- Same CaptureSMB® principles and AutomAb® control at both scales
- Scale-up at over 100× verified and published
- Steady state after one cycle with <1% variability
- YMC ChromaCon provides process transfer support
- FAT — Up to 5 days at YMC facility
- On-site startup — One week including commissioning and system walkdown
- Training — 1 full day or 2 half-days covering operation and maintenance
- Remote support — HMS Ewon remote access for troubleshooting and training
- Warranty: 12 months from installation or 18 months from shipment (whichever first)
- Global service: YMC operations worldwide, LEWA support in 80 countries
- Remote access: Built-in capability for automation troubleshooting
- Components: Non-proprietary with global distribution for local acquisition
- Maintenance: No special tools required for standard maintenance
Yes — customization options include:
- Buffer in-line dilution (BID) with additional pump
- Additional inlet valves (up to 6 on Pump A, B, or C)
- Additional outlet/fraction valves (up to 5 fraction + 1 waste)
- Pre-column analytics (pH and conductivity)
- Single or dual filter housings
- Bubble traps
- CIP manifolds for aseptic single-use bag connection
Contact YMC ChromaCon for custom configuration pricing.
Overview

GMP-Ready Continuous Capture for Biopharmaceutical Manufacturing
The Contichrom® TWIN LPLC – Capture is a sanitary chromatography system for continuous capture of biologics from pilot to large-scale GMP manufacturing.
- Twin-column continuous capture (CaptureSMB® with AutomAb®)
- Up to 3× faster processing, 50% less resin and buffer vs. batch
- Sanitary design with CIP capability for biopharmaceutical production
- SS 316L or Hastelloy flow paths, 7.5 bar maximum operating pressure
- 21 CFR Part 11 compliant software with full audit trail

One System, Multiple Processes
Batch AND continuous capture on a single sanitary skid:
| Mode | Technology |
|---|---|
| Batch | Single-column isocratic and gradient |
| 2D Integrated Batch | Two-step purification with in-line dilution (BID) |
| Continuous Capture | CaptureSMB® with AutomAb® |
- Switch seamlessly between batch and CaptureSMB® modes
- Buffer in-line dilution (BID) option for on-board pH adjustment
- Eliminates duplicate equipment — saves space and capital

Patented CaptureSMB Technology
Twin-column continuous capture maximizing resin binding capacity:
| Metric | Value |
|---|---|
| Speed | Up to 3× faster than batch |
| Resin & buffer | Up to 50% reduction |
| Productivity | 2–4× vs. single-column batch |
- Two columns alternate loading and elution — continuous output
- Columns in series so the second captures breakthrough from the first
- Near-complete resin utilization eliminates re-chromatography

AutomAb® - Dynamic Process Control
AutomAb® monitors UV breakthrough in real time and adjusts column switching automatically:
- Detects product breakthrough and triggers switching at the optimal point
- Compensates for feed variability, column aging, and resin degradation
- Delivers consistent capture efficiency without operator intervention
- Enables robust, continuous 24/7 manufacturing
- Steady state reached after only one cycle with <1% variability
Comprehensive Application Range
Production-scale capture for biopharmaceuticals:
| Application | CaptureSMB® | Integrated Batch |
|---|---|---|
| mAbs & Antibody Variants | ✓ | ✓ |
| Viral Vectors (AAV) | ✓ | ✓ |
| Antibody-drug Conjugates | ✓ | ✓ |
| Recombinant Proteins | ✓ | ✓ |
- All processes run on the same sanitary hardware
- AutomAb® dynamic control across continuous operations
Scalable from Lab to Production
Part of the Contichrom® platform with established transfer protocols:
| System | Flow Rate | Column ID |
|---|---|---|
| CUBE 30/100 | 0.1–100 mL/min | 0.5–5 cm |
| TWIN LPLC 300 | 0.03 – 3.33 L/min | 10–20 cm |
| TWIN LPLC 500 | 0.08 – 8.33 L/min | 20–45 cm |
| TWIN LPLC 1000 | 0.15 – 18.9 L/min | 30–60 cm |
| TWIN LPLC 2000 | 0.5 – 36 L/min | 60–100 cm |
- Scale-up from CUBE at over 100× verified and published
- Same CaptureSMB® and AutomAb® control at every scale
Sanitary Design for Bioprocessing
Purpose-built for biopharmaceutical manufacturing environments:
- Fully sanitary flow path with drainable design and CIP compatibility
- ASME-BPE compliant stainless steel welds on all product contact surfaces
- SS 316L or Hastelloy flow path materials
- SF5 spec product contact tubing (≤ 20 Ra EP)
- Designed for NaOH 1M CIP and compatible with single-use aseptic interfaces
- Maximum operating pressure of 7.5 bar — optimal for affinity and LP resins
Designed for GMP Compliance
Built to meet stringent biopharmaceutical manufacturing requirements:
- Developed under the GAMP 5 framework
- 21 CFR Part 11 compliant software with audit trail
- Comprehensive Turnover Package (TOP) with ASME-BPE compliant welds
- USP Class VI materials and FDA-approved seals
- UL-approved panels, CE marking including PED compliance
TOP includes: MTRs, pressure test reports, passivation reports, weld documentation, as-built drawings.
Production-Grade Hardware
A complete skid-mounted system with mobile frame:
- 2× Pumps (base) — LEWA ecodos triplex with rupture detection
- 3rd Pump (BID option) — Buffer in-line dilution
- Dual UV Detectors — 4-Channel Adjustable wavelength (200-600 nm), 0.5 mm path length
- 2× Coriolis flow meters + 8× pressure transmitters
- pH, conductivity & temperature — Post-column standard
- Air sensors — Pre-pump and pre-column
- Pneumatic valves — Sanitary design, fast-acting at low pressure
- 24″ HMI — Industrial touchscreen with FactoryTalk View SE
CIP-Ready Design
The Contichrom® TWIN LPLC – Capture is designed for cleaning-in-place:
- Dedicated CIP inlet and outlet manifolds integrated into the valve network
- Fully drainable flow path design minimizes carryover between campaigns
- Compatible with NaOH 1M CIP protocols
- Optional single-use aseptic interface for minimally classified suites
- Reduces turnaround time and supports multi-product facility operation
How it works

Sanitary Hardware for GMP Biopharmaceutical Capture Chromatography
The Contichrom TWIN LPLC – Capture is a complete sanitary skid-mounted unit for GMP manufacturing:
- Mobile frame with levelers and non-marking caster wheels
- 316L stainless steel frame with mechanically polished welds
- Sanitary flow path designed for drainability and CIP
- Two column positions for CaptureSMB and single-column batch
- Operating temperature: 4–25 °C ambient, 4–40 °C process
LEWA Ecodos Triplex Pumps
Industry-leading LEWA ecodos triplex pumps for precision and reliability:
| Pump | Role |
|---|---|
| Pump A | Feed delivery / Load buffer |
| Pump B | Elution / Wash / Equilibration |
| Pump C (optional) | Buffer in-line dilution (BID) |
- Triplex configuration for nearly pulse-free flow
- Sandwich diaphragm with rupture detection alarm
- Automatic stroke adjustment for optimal performance
- PTFE diaphragms — USP Class VI certified, FDA approved
- Base system includes 2 pumps; 3rd pump available with BID option
Pneumatic Valve Network
Sanitary pneumatic valves — fast-acting at low pressure:
- Inlet Selection Valves — Multiple ports per pump for automated buffer/feed switching
- Column Switching Valves — For CaptureSMB interconnected and parallel configurations
- Fraction Outlet Valves — Product and waste routing with drain connection
- CIP Manifolds — Dedicated inlet and outlet for cleaning-in-place
- Materials: 316L SS body, PTFE/EPDM diaphragm, SF5 spec product contact (≤ 20 Ra EP)
- Sanitary design
Dual Wavelength UV Detectors
Two post-column UV sensors for real-time monitoring and AutomAb® control:
| Parameter | Specification |
|---|---|
| Type | 4-channel, multi-wavelength DAD, 200–600 nm |
| Path length | 0.5 mm path (exchangeable for 0.1, 0.3, or 1 mm) |
- Provides the UV breakthrough signals that enables AutomAb® dynamic process control
- Critical for consistent CaptureSMB® capture efficiency
In-Line Process Sensors
Comprehensive real-time monitoring:
| Sensor | Quantity | Specification |
|---|---|---|
| Coriolis Flow Meters | 2 | Flow monitoring and control |
| Pressure Transmitters | 8 | 0–13.8 bar, ±1% FS |
| pH | Post-column (x2) | 0–13 pH, ±0.15 unit |
| Conductivity | Post-column (x2) | 0–200 mS/cm, ±1% of measured value |
| Temperature | Per column | 0–100 °C, ±0.25 °C at 25 °C |
| Air Sensors | Pre-pump + pre-column | Ultrasonic detection |
Column Management
Flexible column configurations for production scale:
- 2 column positions for CaptureSMB® twin-column operation
- Column ID range: 10–100 cm (depending on model)
- Low-pressure operation Max 7.5 bar — optimized for affinity resins
- Sanitary tri-clamp connections for column attachment
- Single-column batch mode available
Human-Machine Interface (HMI)
System control via industrial PC running FactoryTalk View SE:
| Component | Specification |
|---|---|
| Touchscreen | 24″ industrial HMI, panel-mounted |
| Industrial PC | Preloaded with control software |
| OS | Windows 11 Professional |
- Real-time process visualization, recipe management, batch reporting
GMP-Grade Flow Path
Every product-contact surface selected for biopharmaceutical compatibility:
| Component | Materials |
|---|---|
| Product Contact Tubing | 316L SS or Hastelloy, ≤ 20 Ra EP (SF5 spec) |
| Non-Product Contact Tubing | 316L SS, ≤ 20 Ra MP (SF1 spec) |
| Process Valves | 316L SS body, PTFE/EPDM diaphragm |
| Pump Diaphragms | PTFE (USP Class VI, FDA approved) |
- ASME-BPE compliant welds, drainable design, CIP-compatible
- Surface roughness < 0.8 µm Ra on wetted metallic parts
PLC-Based Control System
Industrial automation platform:
| Component | Specification |
|---|---|
| PLC Platform | Rockwell CompactLogix |
| HMI Runtime | FactoryTalk View SE |
| Process Control | CaptureSMB with AutomAb® |
| Database | Microsoft SQL Server |
| Historian | YMC Server Historian |
| Reports | YMC Server Reports (auto-generated) |
| Remote Access | HMS Ewon module (client-enabled) |
Optional Hardware
| Option | Specification |
|---|---|
| Buffer In-line Dilution (BID) | Pump C + inlet valves + static mixer + sensors |
| Additional Inlet Valves | Up to 6 per pump |
| Additional Outlet Valves | Up to 5 fraction + 1 waste |
| Filter Housings | Single or dual with valve manifolds |
| Bubble Traps | 2 with valve manifold and vent valves |
| CIP Manifolds | Aseptic single-use bag connection |
| Software Customizations | Handshake with upstream/downstream equipment, additional control loops, external valve/equipment control |
Process Capabilities

Continuous Capture for Biopharmaceutical Production
The Contichrom TWIN LPLC – Capture is optimized for twin-column continuous capture:
- CaptureSMB® with AutomAb® — Continuous capture
- 2D Integrated Batch — Two-step purification with in-line dilution (BID option)
- Batch — Single-column isocratic and gradient operation
- All accessible from a single sanitary GMP-ready platform

CaptureSMB® with AutomAb® — Continuous Capture
Twin-column continuous capture maximizing resin binding capacity:
- Two identical columns alternate between loading and elution
- Columns in series — second column captures breakthrough from the first
- AutomAb® detects UV breakthrough and triggers column switching
- Near-complete utilization of resin binding capacity
- Continuous feed processing eliminates batch size limitations
- Steady state after one cycle with <1% variability
Target: Affinity capture steps (Protein A, mixed-mode) Molecules: mAbs, antibody variants, viral vectors (AAV), ADCs, recombinant proteins
CaptureSMB® — Performance Gains
| Metric | Result |
|---|---|
| Processing speed | Up to 3× faster than batch |
| Protein A resin consumption | Up to 50% reduction |
| Buffer consumption | Up to 50% reduction |
| Productivity | 2–4× higher vs. single-column batch |
| Intermediate storage | Reduced — continuous output eliminates hold steps |
| Process control | AutomAb® enables continuous 24/7 operation |
| Scale-up variability | <1% — steady state after one cycle |
One column loads while the other elutes — continuous output, maximum resin utilization, consistent quality.

Integrated Batch — Two-Step Purification
Couple two chromatography steps without intermediate handling:
- Parallel or in-series column configurations
- Two chromatography modes run consecutively (e.g., IEX then HIC)
- On-board buffer in-line dilution (BID option) for pH adjustment
- Eliminates intermediate hold tanks and manual handling
- Reduces process time, QC burden, and facility footprint
Molecules: mAbs, antibody variants, viral vectors (AAV), ADCs, recombinant proteins
Batch Chromatography
Single-column batch purification:
| Mode | Applications |
|---|---|
| Isocratic | Affinity capture, SEC, desalting |
| Gradient | IEX, HIC, mixed-mode polishing |
- Linear gradient from 5% to 95% in batch mode
- Multiple fraction collection outlets
- Full UV/flow/pressure/pH/conductivity monitoring
Buffer In-line Dilution (BID)
On-board dilution of buffer concentrates and CIP solutions:
- Third pump (Pump C) delivers dilution buffer at controlled ratio
- Static mixer ensures homogeneous blending
- Enables pH adjustment between capture and polish steps
- Supports sequential processing for pool-less multi-step chromatography
Requires: BID option (Pump C, inlet valves, static mixer, sensors)
Regulatory Compliance & Documentation
Certifications
- UL-approved electrical panels
- CE marking available including PED compliance
- Developed under GAMP 5 framework
- Complies with FDA 21 CFR Part 11 regulations
Standards & Directives
- ASME-BPE for all stainless steel product contact welds
- FDA-approved USP Class VI materials and seals
- UL-approved electrical panels
- CE marking with PED compliance
Materials & Documentation
- All wetted materials selected for biopharmaceutical compatibility and chemical resistance
- Full traceability with MTRs and certificates of compliance
- Product contact surface roughness: < 0.8 µm Ra (SF5 spec: ≤ 20 Ra EP)
- Turnover Package (TOP) with comprehensive QA/QC documentation
TOP includes: MTRs, certificates of compliance, pressure test reports, passivation reports, weld documentation (logs, inspection reports, procedures, coupon logs, isometrics), slope verification, operation and maintenance manuals, as-built drawings (PDF and native CAD/Solidworks).
Specifications
System Models
| Parameter | TWIN 300 | TWIN 500 | TWIN 1000 | TWIN 2000 |
|---|---|---|---|---|
| Flow Rate | 0.03 – 3.33 L/min | 0.08 – 8.33 L/min | 0.15 – 18.9 L/min | 0.5 – 36 L/min |
| Column ID | 10–20 cm | 20–45 cm | 30–60 cm | 60–100 cm |
| Max Operating Pressure | 7.5 bar | 7.5 bar | 7.5 bar | 7.5 bar |
| Best For | Pilot / clinical | Mid-scale GMP | Large-scale GMP | Maximum capacity |
General Specifications
| Specification | Value |
|---|---|
| Max Operating Pressure | 7.5 bar (0.75 MPa / 109 psi) |
| Operating Temperature | 4–25 °C ambient; 4–40 °C process |
| Design | Sanitary, CIP-compatible (NaOH 1M) |
| Wetted Materials | SS 316L or Hastelloy, PTFE, EPDM (USP Class VI) |
| Surface Finish | < 0.8 µm Ra (product contact), SF5 spec: ≤ 20 Ra EP |
| Control System | Rockwell CompactLogix PLC |
| Processes | Batch, Integrated Batch, CaptureSMB® |
Physical Specifications
| Model | Length | Width | Height | Approx. Weight |
|---|---|---|---|---|
| TWIN 300 | 218 cm | 127 cm | 203 cm | 2494 kg |
| TWIN 500 | 299 cm | 165 cm | 198 cm | 3055 kg |
| TWIN 1000 (Pump) | 360 cm | 140 cm | 230 cm | 4455 kg |
| TWIN 1000 (Process) | 140 cm | 90 cm | 200 cm | 1364 kg |
| TWIN 2000 (Pump) | 381 cm | 152 cm | 230 cm | 4763 kg |
| TWIN 2000 (Process) | 147 cm | 101 cm | 200 cm | 1543 kg |
Pumps (LEWA ecodos)
| Configuration | 2× triplex diaphragm pumps (3× with BID option) |
| Diaphragm | Sandwich PTFE with rupture detection |
| Materials | USP Class VI certified, FDA approved |
| Features | Automatic stroke adjustment, nearly pulse-free flow |
Detectors & Sensors
| UV Detection | 4-channel, multi-wavelength DAD, 200–600 nm, 0.5 mm path (exchangeable for 0.1, 0.3, or 1 mm) |
| Flow Meters (×2) | Coriolis |
| Pressure (×8) | Transmitters throughout flow path, 0–13.8 bar, ±1% FS |
| pH | On waste and collection outlets, 0–13, ±0.15 unit |
| Conductivity | On waste and collection outlets, 0–200 mS/cm, ±1% of measured value |
| Temperature | Post-column, 0–100 °C, ±0.25 °C at 25 °C |
| Air Sensors | Pre-pump and pre-column (ultrasonic) |
Valves & Flow Path
| Valve Actuation | Pneumatic diaphragm valves |
| Valve Materials | 316L SS body, PTFE/EPDM diaphragm |
| Product Contact | SF5 spec: ≤ 20 Ra EP |
| Non-Product Contact | SF1 spec: ≤ 20 Ra MP |
| Column Connections | Sanitary tri-clamp |
| CIP | Dedicated inlet and outlet manifolds |
Software & Automation
| PLC Platform | Rockwell CompactLogix |
| HMI Runtime | FactoryTalk View SE |
| Process Control | CaptureSMB® with AutomAb® |
| Database | Microsoft SQL Server |
| Historian | YMC Server Historian |
| Reports | YMC Server Reports (auto-generated) |
| Remote Access | HMS Ewon module (client-enabled) |
| Data Integrity | 21 CFR Part 11, audit trail, electronic signatures |
User Interface
| HMI | 24″ industrial touchscreen, panel-mounted |
| Industrial PC | Preloaded with control software |
| OS | Windows 11 Professional |
Optional Modules
| Option | Specification |
|---|---|
| Buffer In-line Dilution (BID) | Pump C + inlet valves + static mixer + sensors |
| Additional Inlet Valves | Up to 6 per pump |
| Additional Outlet Valves | Up to 5 fraction + 1 waste |
| Filter Housings | Single or dual with valve manifolds |
| Bubble Traps | 2 with valve manifold and vent valves |
| CIP Manifolds | Aseptic single-use bag connection |
| Software Customizations | Handshake with upstream/downstream equipment, additional control loops, external valve/equipment control |
Technologies

Single-column purification with full method flexibility — gradient, isocratic, step elution. The foundation for process development and the starting point for continuous workflows.
Single-column purification with full method flexibility — gradient, isocratic, step elution. The foundation for process development and the starting point for continuous workflows.

Automate sequential purification across multiple columns and chemistries in a single run. ChromIQ® handles fraction transfer, timing, and method switching.
Automate sequential purification across multiple columns and chemistries in a single run. ChromIQ® handles fraction transfer, timing, and method switching.

Twin-column continuous loading maximizes resin utilization and throughput. AutomAb® dynamically controls breakthrough — adapting in real time to feed variability.
Twin-column continuous loading maximizes resin utilization and throughput. AutomAb® dynamically controls breakthrough — adapting in real time to feed variability.

Real-time UV-based control that adapts your continuous process to feed and column variability — eliminating the need for fixed, conservative operating parameters.
Real-time UV-based control that adapts your continuous process to feed and column variability — eliminating the need for fixed, conservative operating parameters.
Applications

CaptureSMB® with AutomAb® for mAbs & antibody variants: maximize productivity and resin utilization, while slashing buffer consumption.
CaptureSMB® with AutomAb® for mAbs & antibody variants: maximize productivity and resin utilization, while slashing buffer consumption.

From capture through polishing, Contichrom® integrates multiple chromatography steps into automated 2D/3D workflows — reducing hands-on time and intermediate handling.
From capture through polishing, Contichrom® integrates multiple chromatography steps into automated 2D/3D workflows — reducing hands-on time and intermediate handling.

MCSGP with AutoPeak® is ideally suited for DAR control, achieving precise drug-antibody ratios with higher yield than batch chromatography.
MCSGP with AutoPeak® is ideally suited for DAR control, achieving precise drug-antibody ratios with higher yield than batch chromatography.
Resources
- All Media Types
- Peer-Reviewed Publications
Continued insights into virus clearance validation across continuous capture chromatography, Biotechnol. Bioeng.
Additional viral validation studies for CaptureSMB validate a surrogate batch model that accurately predicts virus clearance in continuous capture operations.
Model-based process development of continuous chromatography for antibody capture: A case study with twin-column system, J. Chromatogr. A
Model-based approaches for twin-column continuous capture process design establish systematic methodology for CaptureSMB implementation.
Virus Clearance Validation across Continuous Capture Chromatography. Biotechnol. Bioeng.
Viral validation studies conducted under cGLP guidelines for CaptureSMB demonstrate comparable virus clearance to batch processing and establish regulatory-compliant validation approaches.
Service & Support

See Contichrom® in action. On-site, at our facility, or remote — get a first-hand look at what our chromatography systems and software can do.
See Contichrom® in action. On-site, at our facility, or remote — get a first-hand look at what our chromatography systems and software can do.

Hands-on operator training, advanced application workshops, and expert consulting — customised to your team's experience and your specific Contichrom® applications.
Hands-on operator training, advanced application workshops, and expert consulting — customised to your team's experience and your specific Contichrom® applications.

Calibrated mechanistic models built from your batch data — simulate and optimize batch, MCSGP, or CaptureSMB® processes computationally, before running a single experiment.
Calibrated mechanistic models built from your batch data — simulate and optimize batch, MCSGP, or CaptureSMB® processes computationally, before running a single experiment.

Try a Contichrom® system in your own lab before committing to a purchase. Full technical support and training included — evaluate the technology on your terms.
Try a Contichrom® system in your own lab before committing to a purchase. Full technical support and training included — evaluate the technology on your terms.

Quantified performance comparison. Run your molecule on our Contichrom® systems and benchmark CaptureSMB® or MCSGP against your existing batch process.
Quantified performance comparison. Run your molecule on our Contichrom® systems and benchmark CaptureSMB® or MCSGP against your existing batch process.

Preventive maintenance subscriptions and on-site repair services to keep your Contichrom® running at peak performance. Remote technical support available worldwide.
Preventive maintenance subscriptions and on-site repair services to keep your Contichrom® running at peak performance. Remote technical support available worldwide.
FAQ
A complete Contichrom® TWIN LPLC – Capture system includes:
- Complete sanitary skid assembly with mobile frame (levelers and casters)
- 2× LEWA ecodos triplex pumps with sandwich diaphragms and rupture detection
- Full inlet and outlet pneumatic valve network (sanitary design)
- Dual wavelength UV detectors (280 nm, 303 nm)
- Post-column pH and conductivity sensors
- 2× Coriolis flow meters + 8× pressure transmitters
- Pre-pump and pre-column air sensors
- CIP inlet and outlet manifolds
- 24″ HMI with industrial PC and control software preloaded
- Rockwell CompactLogix PLC control system
- Comprehensive TOP documentation package
- Factory Acceptance Testing (FAT) — up to 5 days
- One week on-site startup support and training
Customer provides: Columns, filter elements, chromatography resin, external piping, utilities connections.
| Feature | TWIN HPLC | TWIN LPLC Capture | TWIN LPLC Polishing |
|---|---|---|---|
| Design | Industrial (non-sanitary) | Sanitary | Sanitary |
| Pressure | 80 bar | 7.5 bar (20 bar opt.) | 7.5 bar (20 bar opt.) |
| Core Process | MCSGP (polishing) | CaptureSMB® (capture) | MCSGP (polishing) |
| HP Valves | YMC fast-acting | Standard pneumatic | Standard pneumatic |
| Pumps | 4× LEWA ecoflow | 2× (3×) LEWA ecodos | 4× LEWA ecodos |
| Molecules | Synthetic peptides, oligos | Biologics (mAbs, AAV, ADCs) | Biologics (mAbs, AAV, ADCs) |
The TWIN LPLC – Capture is the right choice for affinity capture of biopharmaceuticals where sanitary design and CIP capability are required.
| Model | Column ID | Flow Rate | Best For |
|---|---|---|---|
| TWIN 300 | 10–20 cm | 0.03 – 3.33 L/min | Pilot production, clinical manufacturing |
| TWIN 500 | 20–45 cm | 0.08 – 8.33 L/min | Mid-scale commercial production |
| TWIN 1000 | 30–60 cm | 0.15 – 18.9 L/min | Large-scale commercial production |
| TWIN 2000 | 60–100 cm | 0.5 – 36 L/min | Maximum capacity production |
Contact YMC to discuss your throughput requirements and optimal system selection.
- Floor space: Dimensions vary by model (contact YMC for GA drawings)
- Power: Model-dependent (contact YMC for specifications)
- Utilities: Process air, water, compressed air, drain, network connection
- Ambient temperature: 4–25 °C
- Process temperature: 4–40 °C
Note: System must be uncrated, leveled, and connected to utilities before YMC on-site work begins.
- Methods developed on Contichrom CUBE transfer directly to the TWIN LPLC
- Same CaptureSMB® principles and AutomAb® control at both scales
- Scale-up at over 100× verified and published
- Steady state after one cycle with <1% variability
- YMC ChromaCon provides process transfer support
- FAT — Up to 5 days at YMC facility
- On-site startup — One week including commissioning and system walkdown
- Training — 1 full day or 2 half-days covering operation and maintenance
- Remote support — HMS Ewon remote access for troubleshooting and training
- Warranty: 12 months from installation or 18 months from shipment (whichever first)
- Global service: YMC operations worldwide, LEWA support in 80 countries
- Remote access: Built-in capability for automation troubleshooting
- Components: Non-proprietary with global distribution for local acquisition
- Maintenance: No special tools required for standard maintenance
Yes — customization options include:
- Buffer in-line dilution (BID) with additional pump
- Additional inlet valves (up to 6 on Pump A, B, or C)
- Additional outlet/fraction valves (up to 5 fraction + 1 waste)
- Pre-column analytics (pH and conductivity)
- Single or dual filter housings
- Bubble traps
- CIP manifolds for aseptic single-use bag connection
Contact YMC ChromaCon for custom configuration pricing.



